Enzyme activity as an actionable axis for small-molecule precision oncology
Enzyme activity as an actionable axis for small-molecule precision oncology
Fujita, K.; Kamiya, M.; Dan, S.; Tachibana, R.; Kawatani, M.; Kojima, R.; Hino, R.; Kobayashi, K.; Inoue, S.; Tani, M.; Hirata, Y.; Kawashima, S.; Yamazaki, K.; Nishimura, Y.; Ohashi, Y.; Isoyama, S.; Nakada, A.; Matsumoto, N.; Ikegaya, Y.; Nakajima, J.; Urano, Y.
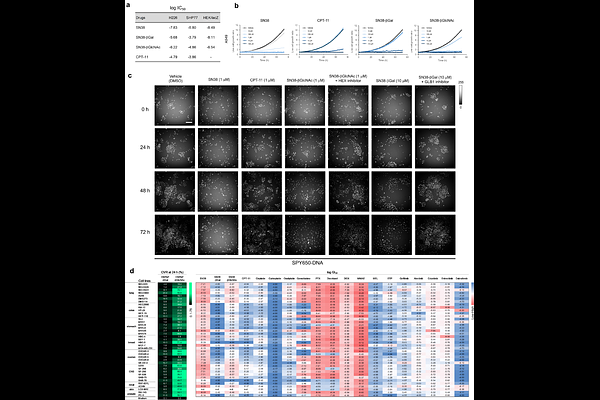
AbstractCurrent precision oncology--molecular targeted therapies and immunotherapies--relies on genomic or expressed biomarkers, yet most cancer patients remain ineligible for these treatments. Here, we establish enzyme activity as an actionable and orthogonal axis for precision cancer medicine. Strategic activity-based screening of mouse organs and human clinical specimens with a panel of enzyme-reactive fluorescence probes identified {beta}-galactosidase 1 (GLB1) and {beta}-hexosaminidases (HEX) as broadly elevated tumor-selective biomarkers. Leveraging these activities, we developed 7-ethyl-10-hydroxycamptothecin (SN38)-based GLB1- and HEX-reactive prodrugs. These prodrugs exhibited dramatically reduced systemic toxicities and improved therapeutic windows, compared to a clinically used SN38-based prodrug, irinotecan (CPT-11). Both prodrugs demonstrated activity-dependent therapeutic efficacy, affording a dramatic reduction of tumor volumes across multiple in vivo models, including a subcutaneous patient-derived xenograft (PDX) of lung squamous cell carcinoma that lacked genetic alterations targeted by current precision medicine. Furthermore, this strategy is broadly applicable across various cytotoxic payloads, establishing a generalizable platform for small-molecule precision medicines. Our results define an enzyme-targeting paradigm for precision oncology, in which fluorescence probes serve as companion diagnostic tools to guide development and selection of appropriately targeted prodrugs, which are expected to provide safer and more efficacious treatment options for cancer patients with elevated enzyme activities.