Human PARPs modify RNA nucleobases in vitro and in cells
Human PARPs modify RNA nucleobases in vitro and in cells
Musheev, M. U.; Siefert, J.; Bauten, W.; Butepage, M.; Luscher, B.; Niehrs, C.; Feijs, K. L.; Zaja, R.
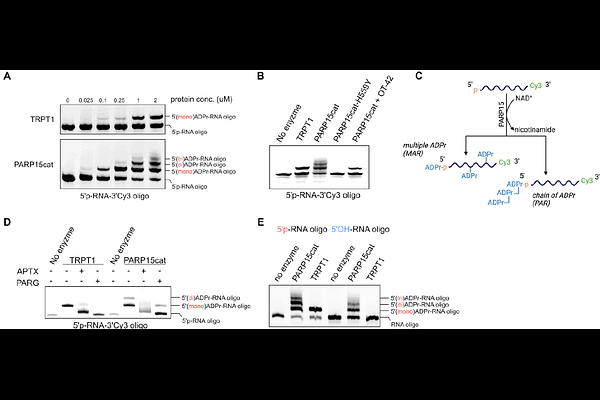
AbstractADP-ribosylation is known as a protein modification, yet recent studies have expanded the range of ADP-ribosyltransferase (ART) substrates to include nucleic acids. tRNA 2'-phosphotransferase 1 (TRPT1) and several PARP family members can modify the 5'-phosphate of single-stranded RNA. Here, we show that PARP10 and PARP15 extend this activity beyond the 5'-phosphate terminus and generate N3-ADP-ribosyl uracil and N1-ADP-ribosyl guanine bases. The base-linked ADP-ribosylation is reversed selectively by the macrodomain-containing hydrolase TARG1. In TARG1 knockout cells, N1-ADP-ribosyl guanine can be detected. Together, these findings establish guanine and uracil ADP-ribose as two novel nucleotide modifications and reveal PARP15 and TARG1 as an enzyme pair which can dynamically regulate guanine ADP-ribosylation in living cells.