Denatured Albumin Gains a Function of Regulating Platelet Activity
Denatured Albumin Gains a Function of Regulating Platelet Activity
Pandey, V.; Kundu, S.; Wang, S.; Pyne, A.; Que, L.; Wang, X.
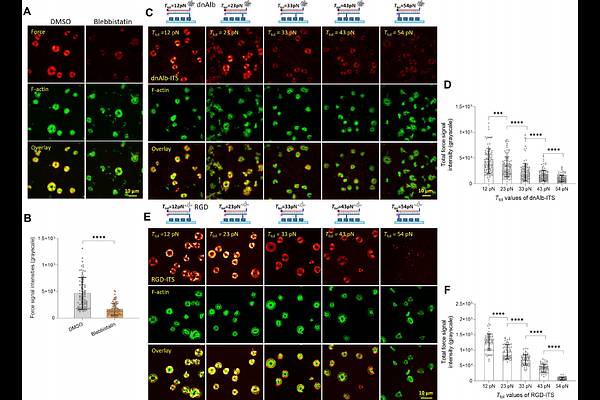
AbstractPlatelets are blood cells that play a critical role in hemostasis and thrombosis. Serum albumin, which constitutes approximately 50% of plasma proteins, has traditionally been considered non-interactive with platelets and not involved in platelet function. Here, using a molecular force sensor and platelet adhesion and aggregation assays, we show that serum albumin, if denatured, specifically binds to integrins in platelets and transmits platelet contractile forces, regulating platelet activation, adhesion and aggregation as effectively as fibrinogen (the clotting factor I). 0.1% ethanol or 10 micromolar hydrogen peroxide, physiologically attainable in blood through alcohol intake or disease-elevated oxidative stress, are sufficient to denature albumin into a functional platelet ligand. These findings revealed albumin as a previously unrecognized but important regulator of platelet functions, with broad implications for the thrombotic risk in the context of physiological conditions that induce albumin denaturation.