NFATC2 in pancreatic cancer-associated fibroblasts predicts treatment response and facilitates ERBB-targeted therapies
NFATC2 in pancreatic cancer-associated fibroblasts predicts treatment response and facilitates ERBB-targeted therapies
GUO, J.; Cancellieri, S.; Xu, C.; Wiik, C.; Fei, L.; Dahal-Koirala, S.; Haapaniemi, E.; Aittokallio, T.; Verbeke, C. S.; Sahu, B.
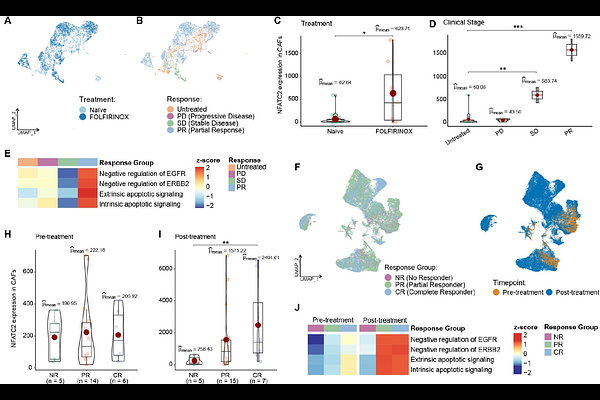
AbstractPancreatic ductal adenocarcinoma (PDAC) remains a lethal malignancy, with therapeutic resistance influenced by a dense desmoplastic stroma dominated by cancer-associated fibroblasts (CAF). Using single-cell RNA-sequencing and gene regulatory network modeling of 42 PDAC tumors, we identified a CAF subpopulation characterized by elevated NFATC2 expression that is enriched in patients with improved therapeutic response and survival. NFATC2+ CAFs exhibited tumor-suppressive features, including enhanced apoptotic signaling and suppression of ERBB pathway activity. Co-culture experiments demonstrated that NFATC2+ CAFs restrain pancreatic cancer cell growth and enhance chemotherapy-induced apoptosis, increasing sensitivity to standard-of-care chemotherapy regimens and synergizing with ERBB-targeted therapies. The favorable effect of NFATC2+ CAFs on chemotherapy response was validated in two other PDAC cohorts and in rectal cancer. Together, these findings identify NFATC2+ CAFs as a therapy-conditioned stromal state linked to improved treatment response and uncover a context-dependent vulnerability within the tumor microenvironment that may be exploited to rationally optimize combination therapies.