Disrupting miR-466l-3p and HuR Cooperation with Target Site Blockers Reveals a Therapeutic Strategy to Destabilize mRNA Transcripts
Disrupting miR-466l-3p and HuR Cooperation with Target Site Blockers Reveals a Therapeutic Strategy to Destabilize mRNA Transcripts
Ramgolam, V.; Yarovinsky, T. O.; Huntenburg, S.; Bergman, C.; Ruddle, N.; Bender, J.
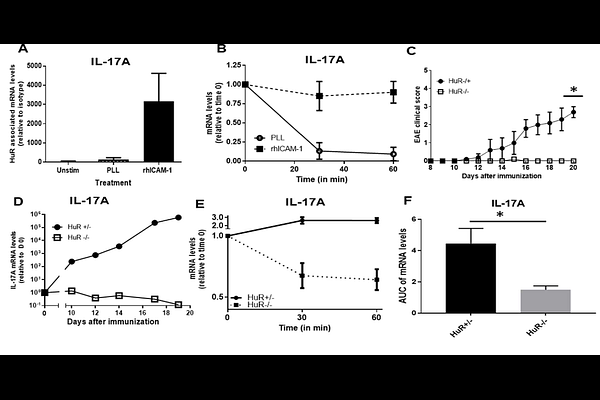
AbstractMicroRNAs (miRNAs) typically regulate gene expression by promoting mRNA degradation, but select miRNAs, such as miR-466l-3p (miR-466), can instead stabilize transcripts in coordination with RNA-binding proteins (RBPs) like HuR. We identify conserved AU-rich elements (cAREs) within the 3'-UTRs of IL-17A, GM-CSF, and IL-23A as critical cis-regulatory binding site where miR-466 facilitates HuR recruitment to promote mRNA stability. Using site-directed mutagenesis, RNA pulldown, and MS2-TRAP assays to capture miRNA-mRNA complexes, we demonstrate that HuR binding depends on prior engagement by miR-466. Disrupting this interaction with rationally designed Target Site Blockers (TSBs) destabilizes target mRNAs and suppresses cytokine expression in vitro and in vivo. TSBs directed against IL-17A, GM-CSF, and IL-23A selectively blocked miR-466 binding, reduced transcript stability, and lowered cytokine production without affecting unrelated mRNAs. In murine models of LPS-induced inflammation, psoriasis, and autoimmunity, TSBs exhibited therapeutic efficacy and cytokine specificity, outperforming monoclonal antibodies in some settings. Phosphorothioate-modified TSBs enabled systemic delivery and retained activity in human T cells, underscoring translational potential. Similar to antisense oligonucleotides, TSBs trigger RNase H1-mediated degradation while also blocking miRNA-mRNA interactions. These findings establish miR--66-HuR cooperation as a therapeutically targetable axis through TSBs without affecting global miRNA function.