rRNA Expansion Segments Mediate Ribosome Dimerization as a Conserved Stress Response
rRNA Expansion Segments Mediate Ribosome Dimerization as a Conserved Stress Response
Jiang, W.; Chen, C.; Wang, X.; Huang, W.; Taylor, D.; Chen, Z.; Xie, J.; Su, Z.; Taylor, D.; Hatzoglou, M.; Guo, Q.
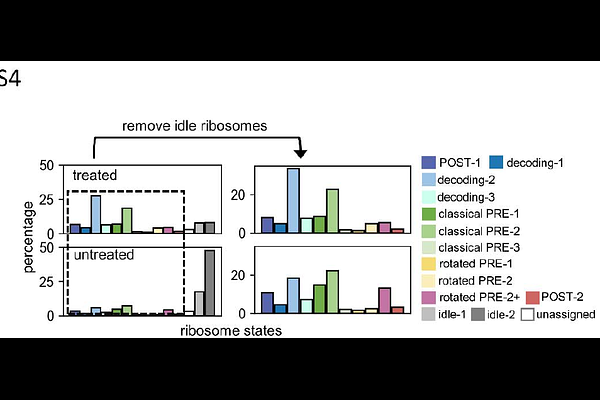
AbstractInhibition of mRNA translation is a common feature in proteostatic stress cellular responses. Puromycin, a widely used compound for studying translation, disrupts protein synthesis by mimicking the 3' end of aminoacyl-tRNAs. Despite its extensive use as a research tool to probe the connection between translation activity and various physiological and pathological states, the cellular response associated with puromycin-induced translation stress remains incompletely understood. Utilizing in situ cellular electron tomography and topology analysis, we visualized the translation machinery at high resolution. Our analysis revealed that puromycin-treated neuronal cells exhibit an accumulation of "idle ribosomes" characterized by the binding of eIF5A, indicating a close association of this factor with translationally inactive ribosome states under stress. Additionally, the idle ribosomes formed dimeric complexes mediated by ribosomal RNA expansion segments, suggesting an evolved mechanism involving these regions in translation hibernating and protecting idle ribosomes. We show that the hibernating disome formation is not unique to puromycin administration but represents a conserved mechanism as a response to different cellular stressors including those associated with endoplasmic reticulum (ER) stress and amino acid depletion. Altogether, our findings shed light on previously unexplored aspects regarding unique states of mammalian ribosome hibernation, and collectively offers new avenues for understanding the correlation of cellular stress response and the regulation of protein synthesis.