Small molecule ensembles reshape amyloid aggregation landscapes
Small molecule ensembles reshape amyloid aggregation landscapes
Nair, R. V.; Tran, B. N.; Parikh, A. N.; Foreman, M. R.
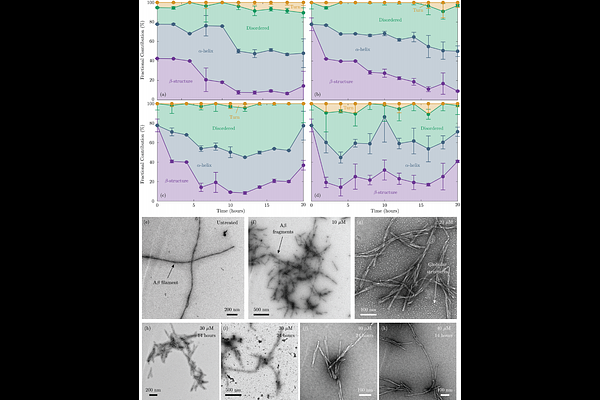
AbstractAmyloid-{beta}42 (A{beta}42) assemblies form a dynamic network of oligomers and fibrils, with fibrillar species acting as reservoirs that maintain equilibrium among intermediates. Perturbing a single species shifts the oligomer-fibril balance, highlighting the challenge of selectively targeting toxic species while maintaining the dynamic equilibrium of the amyloid network. Here, we show that the small molecule EPPS (4-(2-hydroxyethyl)-1-piperazine-propanesulfonic acid) fine-tunes this network through cooperative, concentration-dependent disaggregation. At optimal concentrations, EPPS efficiently shifts the equilibrium away from the fibrillar structures via multisite, allosteric interactions. At higher concentrations, EPPS self-assembles into supramolecular clusters, depleting free molecules and allowing partially disaggregated amyloid intermediates to reassemble. Notably, at elevated concentrations, interactions transition from molecule-to-molecule to higher-order ensemble-to-ensemble engagement, where EPPS clusters and amyloid fibrils mutually reshape each other's dynamics. Molecular crowding, modeled with polyethylene glycol, further restricts EPPS access to fibrillar surfaces, modulating activity. These findings reveal that small-molecule dynamics-including cooperative binding, self-assembly, and environment-dependent accessibility-critically govern amyloid network control, providing a mechanistic blueprint for rational design of next-generation amyloid-targeting therapeutics.