Sex differences in osteoblast matrix maturation regulate osteoblast-endothelial interactions
Sex differences in osteoblast matrix maturation regulate osteoblast-endothelial interactions
Sharma, A.; Emery, R.; Pitsillides, A. A.; Clarkin, C. E.
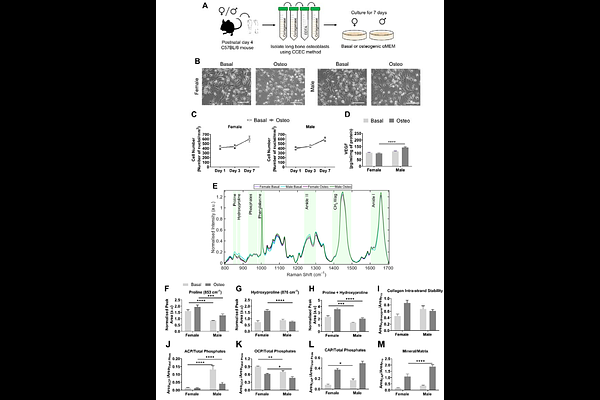
AbstractBackground: Bone formation during development and repair is divergently modulated by osteoblast (OB)-derived vascular endothelial growth factor (VEGF) which drives the skeletal sexual dimorphism of the bone vasculature. While the extracellular matrix (ECM) provides both structural and instructive cues to developing vasculature, the contributions of the bone matrix to this skeletal vascular dimorphism in bone remains undefined at the cellular level. Methods: Primary OBs were isolated from neonatal female and male C57BL/6 long bones and cultured under basal or osteogenic conditions. ECM composition was quantified by Raman spectroscopy. Primary murine bone marrow endothelial cells (BMECs) were seeded directly onto established OB layers and maintained in heterotypic cocultures to assess contact-mediated effects of OB ECM on BMEC survival and expansion. OB-conditioned media (CM) was used to evaluate soluble-factor contributions, with VEGF-A concentration quantified by ELISA. Results: Raman spectroscopy, on individual OBs from monotypic cultures, revealed sexually dimorphic ECM signatures that were independent of cellular growth profiles. Female OB matrices were enriched with type I collagen-specific proline and hydroxyproline and octacalcium phosphate with enhanced collagen intra-strand stability consistent with a matrix-dominant signature. Male OB matrices exhibited relatively lower type I collagen content and higher carbonated apatite resulting in an elevated mineral-to-matrix ratio indicative of advanced mineral maturation. After 24-hours of heterotypic culture of BMECs with OBs, BMEC numbers were 1.39-fold higher when in contact with male OBs. CM treatment of BMECs did not recapitulate these effects despite higher VEGF-A release from male OBs. Conclusions: Sex differences in OB ECM are linked to divergent, contact-dependent regulation of BMEC behaviour. These findings suggest that differences in matrix maturation stat contribute to the sex-specific regulation of the skeletal vascular niche. Elucidating the mechanisms that regulate sex-specific OB-ECM production may reveal new therapeutic targets for selectively modulating pathological skeletal angiogenesis in men and women.