Harnessing NCX-IP3R-dependent Calcium Oscillations to Regulate Angiogenic Signaling in Endothelial Cells
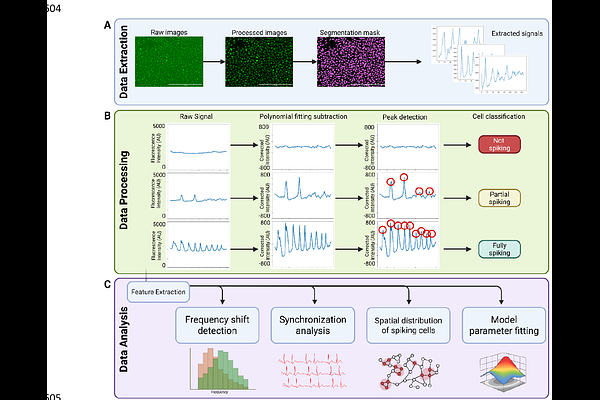
Harnessing NCX-IP3R-dependent Calcium Oscillations to Regulate Angiogenic Signaling in Endothelial Cells
Jeong, D. P.; Cini, S.; Mendiola, K.; Senapati, S.; Dowling, A.; Chang, H.-C.; Zartman, J. J.; Hanjaya-Putra, D.
AbstractThe blood vasculature has a high capacity for structural regeneration, driven by the blood endothelial cells (BECs) that comprise it. This regenerative process, which involves BEC migration and proliferation to form these complex tissues, is linked to low frequency (< 0.1 Hz) calcium spiking that precedes these activities. However, we need new approaches to stimulating angiogenic responses in tissue engineering applications. By conducting experiments that manipulate local ionic concentrations and developing a simple, yet powerful, computational analysis, we demonstrate that sodium-calcium cross-talk is a crucial component that regulates the calcium signaling and downstream angiogenic responses. Activation and deactivation of the inositol triphosphate 3 receptors (IP3Rs) on the endoplasmic reticulum (ER) and the switch between forward and reverse modes of the sodium-calcium exchanger (NCX) are proposed to be the key mechanisms underlying calcium oscillations when cells are exposed to temporary cationic depletion. The spiking is suggested to be a release of intracellular calcium mediated by IP3R activity, and transport in or out of the cell is driven by NCX for the calcium oscillatory signaling pattern. The NCX and IP3R both contribute to manage intracellular calcium and ionic concentration as initially there is a long ER deactivation period while intracellular sodium slowly increases until a sudden onset of calcium is released by the ER. Other calcium and sodium ion channels can change this resonant coupling of ER and NCX to alter the inter-spike duration. Synchronization of the spiking intervals between cells is triggered by stimulating with vascular endothelial growth factor (VEGF), which induces a propagating wave of intracellular calcium across the 2D tissue culture, prior to coordinated cell migration and proliferation towards the VEGF source. This wave, which can be artificially induced and studied using electrical stimulation, suggests that the underlying sodium-calcium crosstalk mechanism introduces intracellular calcium polarization, whose orientation is transferred across cells through spike synchronization. Thus, control of calcium signaling dynamics through regulation of ionic depletion can serve as useful method for generating angiogenic responses in engineered tissue constructs.