c-Fos enhances influenza virus replication by stabilizing the M2 protein and promoting autophagosome accumulation
c-Fos enhances influenza virus replication by stabilizing the M2 protein and promoting autophagosome accumulation
Chen, X.; Yan, J.; Li, M.; Liu, H.; Lu, D.-q.; Wang, Y.; Zhang, Q.; Gao, F.; Peng, J.
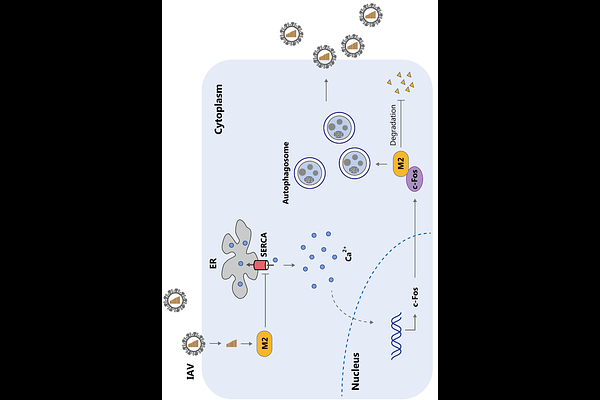
AbstractThe influenza A virus (IAV) employs multiple strategies to hijack host cellular machinery for efficient replication. While autophagosome accumulation is known to promote IAV replication, and the viral matrix protein 2 (M2) ion channel protein plays essential roles in viral uncoating, assembly, and autophagy induction, the mechanisms regulating M2 stability remain incompletely understood. Furthermore, although c-Fos participates in the replication of various viruses, its function in IAV infection has not been characterized. Here, we identify c-Fos as a critical proviral host factor that enhances IAV replication through stabilizing M2 protein and promoting autophagosome accumulation. We demonstrate that IAV infection triggers M2-mediated cytosolic calcium elevation, which upregulates c-Fos expression. The induced c-Fos physically interacts with M2 and prevents its proteasome and lysosomal degradation, thereby increasing M2 accumulation. This stabilized M2 protein as a viral protein directly facilitates viral replication while simultaneously promoting autophagosome accumulation to further enhance the process. Our findings reveal a novel positive feedback loop in IAV infection, establishing c-Fos as a key regulator of IAV replication through M2 stabilization, and highlighting these interactions as potential therapeutic targets for antiviral intervention.