Endosomal maturation is controlled by the trimeric Bulli-Mon1-Ccz1 GEF7 complex and Rab5-GTPase activating protein GAPsec
Endosomal maturation is controlled by the trimeric Bulli-Mon1-Ccz1 GEF7 complex and Rab5-GTPase activating protein GAPsec
Janz, M.; Drechsler, M.; Meyer, H.; Sriram, V.; Simes, K. M.; Frommhold, E.; Füllbrunn, N.; Langemeyer, L.; Ungermann, C.; Kümmel, D.; Paululat, A.
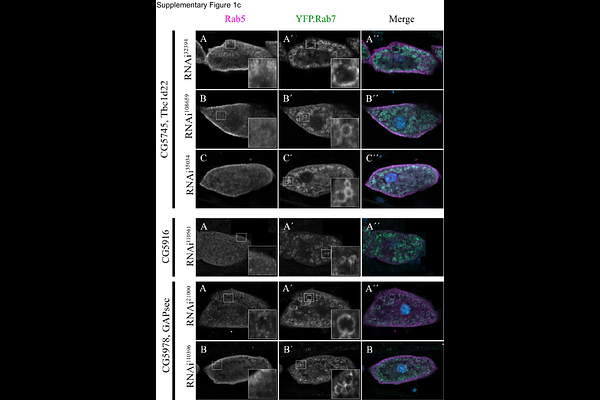
AbstractThe endolysosomal system is crucial for the degradation of cellular waste in the lysosomal lumen. Within this pathway, endosomes mature prior to their fusion with lysosomes. This process relies on the sequential action of the CORVET and HOPS tethering complexes, guided by Rab5 and Rab7 GTPases, respectively. CORVET acts on early endosomes (EEs), transitioning to HOPS on maturing late endosomes/multivesicular bodies (LEs/MVBs) for lysosomal fusion. This process is finely tuned by the Rab activating guanine nucleotide exchange factor (GEF) and the inactivating GTPase activating protein (GAP). The BuMC1 GEF complex (Bulli-Mon1- Ccz1) uniquely activates Rab7 in metazoans and interacts with Rab5, which stimulates its activity. Here, we identified GAPsec as a novel GAP with activity for Rab5 required for endosomal maturation in fruit fly nephrocytes. Inactivation of GAPsec results in enlarged, dysfunctional endosomes that are unable to reach lysosomes for degradation. Our study highlights the importance of coordinated Rab regulation for efficient endosomal trafficking.