Collective microfibril sliding underlies plant cell wall creep
Collective microfibril sliding underlies plant cell wall creep
Li, C.; Yu, J.; Hsia, K. J.; Cosgrove, D. J.; Zhang, S.
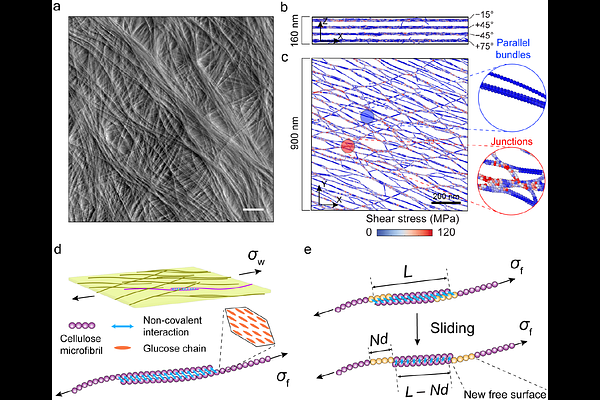
AbstractPlant cell growth depends on slow, irreversible creep of the fibrous cell wall stretched by turgor, yet the mechanics of creep--and how it differs from elasto-plastic deformation--remains uncertain. Using multiscale modeling, we show how wall creep emerges from elementary sliding of cellulose microfibrils along their contact interfaces. A dominant sliding mode involves localized dislocation-like defects that stochastically nucleate at laterally bonded microfibril contacts and then glide along the interface by surmounting discrete energy barriers. Embedding these sliding events into Monte Carlo simulations of stretched cellulose networks recapitulates whole-wall creep kinetics. Elevated stress lowers sliding energy barriers and triggers rapid irreversible deformation characteristic of plastic flow. Compared with elasto-plastic stretching, creep induces less microfibril reorientation but promotes bundling, thereby reducing subsequent yielding. These results clarify the microfibril rearrangements underlying cell wall creep and show how they redistribute stress to permit sustained wall expansion at steady turgor without compromising structural integrity.