Atf3 Integrates Lipid and Cytoskeletal Remodeling to Drive Macrophage Fusion
Atf3 Integrates Lipid and Cytoskeletal Remodeling to Drive Macrophage Fusion
Correia, A.; Mroueh, N.; Wollert, E. K.; Stankovic, D.; Csordas, G.; Juengst, C.; Tartiere, A. G.-B.; Gomes, M.; Iden, S.; Uhlirova, M.
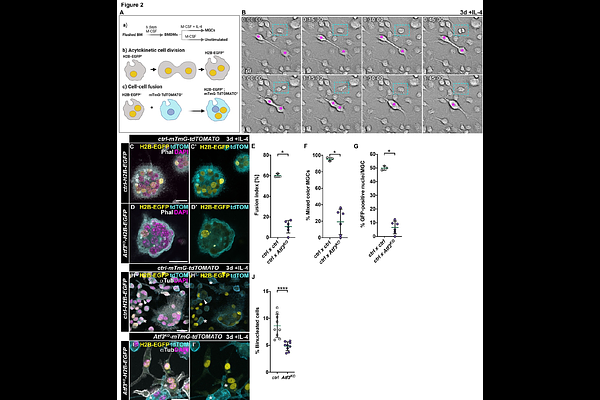
AbstractMacrophages are highly plastic innate immune cells that can form multinucleated giant cells (MGCs) under physiological and pathological conditions, such as osteoclasts and foreign body giant cells. The mechanisms governing macrophage multinucleation remain incompletely understood. Here, we identify activating transcription factor 3 (Atf3) as an essential regulator of MGC formation via cell-cell fusion in response to persistent Interleukin 4 (IL-4) and STAT6 signaling, characteristic of the foreign body reaction. Atf3-deficient macrophages activate STAT6-dependent transcriptional programs in response to IL-4, including fusion-associated genes, yet fail to undergo multinucleation. This defect is associated with impaired actin cytoskeleton remodeling, abnormal nuclear morphology, reduced lamin A/C expression, and genome instability. Mechanistically, loss of Atf3 derepresses the Cholesterol-25-hydroxylase (Ch25h), elevating 25-hydroxycholesterol (25-HC), suppressing the mevalonate pathway, and reducing cholesterol and isoprenoid biosynthesis essential for cytoskeletal dynamics. Deletion of Ch25h in Atf3-deficient macrophages restores cholesterol levels and actin turnover, but not lamin A/C or fusion. These findings establish Atf3 as a central transcriptional node integrating lipid metabolism with cytoskeletal and nucleoskeletal remodeling to control IL-4-driven macrophage multinucleation.