A mechanistic model of protein kinase A dynamics under pro- and anti-nociceptive inputs
A mechanistic model of protein kinase A dynamics under pro- and anti-nociceptive inputs
Lakrisenko, P.; Isensee, J.; Hucho, T.; Weindl, D.; Hasenauer, J.
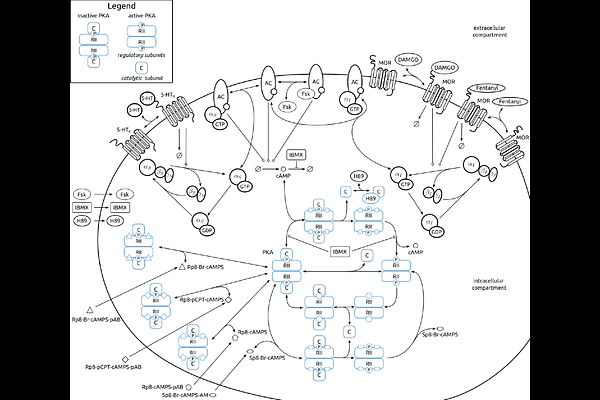
AbstractProtein kinase A (PKA) is a central integrator of nociceptive signaling, yet a quantitative account of how pro- and anti-nociceptive inputs shape its dynamics remains incomplete. Here, we develop a mechanistic model of PKA activity in nociceptive neurons that explicitly links receptor activation to downstream kinase regulation. Using time-course and dose-response measurements, we infer unknown process parameters and quantify parameter and prediction uncertainties to ensure robust conclusions. The model captures the activation of PKA by serotonin and forskolin and its suppression by opioids. We show how the model can be used for the assessment of alternative circuit topologies, and demonstrate that receptor context and stimulation history reconfigure PKA responsiveness, providing testable predictions for opioid modulation under clinically relevant dosing. This framework offers a principled basis for integrating PKA with broader pain-signaling networks, supports rational exploration of combination therapies, and establishes a general strategy for disentangling neuromodulatory control of kinase activity.