Quantitative and mutational analysis of soluble HIV-1 Vpu and calmodulin interactions
Quantitative and mutational analysis of soluble HIV-1 Vpu and calmodulin interactions
Ogunbowale, A.; Hadadianpour, E.; Ishola, O.; Islam, M. M.; Ramos, N.; Saffarian Delkhosh, A.; Georgieva, E. R.
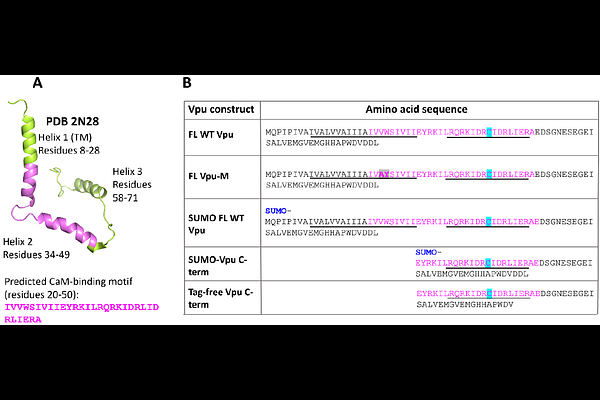
AbstractThe HIV-1 Vpu protein aids viral adaptation by influencing host cell pathways via protein interactions. While Vpu is mainly found in plasma and endomembranes, we recently discovered a soluble form that forms a stable, equimolar complex with Ca2+-bound calmodulin (Ca2+-CaM), potentially affecting Vpu cellular trafficking. Here, to determine the binding affinity and identify regions of soluble Vpu involved in CaM binding, we used ensemble Forster Resonance Energy Transfer (eFRET). We tested Cy3-labeled full-length (FL) Vpu, a C-terminal fragment (helices 2 and 3), and a Cy3-labeled FL Vpu V22A/W23Y mutant with substitutions in Vpu helix 1. All Vpu variants were labeled at residue L42C, and Ca2+-CaM was tagged with Cy5 at residue S39C. eFRET analysis of 100 nM Cy3-Vpu variants mixed with Cy5-Ca2+-CaM (in the range 100-600 nM) revealed dissociation constants (Kd) and binding energies (deltaG) for heterocomplexes. FL Vpu-Ca2+-CaM showed high stability (Kd ~40 nM, deltaG ~10.1 kcal/mol), while the truncated C-terminal region and V22A/W23Y mutant formed less stable complexes with Ca2+-CaM (Kd ~200 nM and 800 nM, deltaG ~9 kcal/mol and ~8.3 kcal/mol). This, a binding hot spot in Vpu CaM-binding motif in helix 1 was identified, which may control the stability of Vpu-Ca2+-CaM complex and Vpu insertion in the membrane: We hypothesize that upon delivery to the membrane, the hydrophobic helix 1 of Vpu dissociates from Ca2+-CaM and inserts in the lipid bilayer; thereafter, CaM dissociates from Vpu facilitated by the reduced Vpu-Ca2+-CaM complex stability. The findings from this study advance our understanding of HIV-1 Vpu interactions with cellular components and may aid the development of antivirals.