UFD1 Recognition of Initiator and Proximal Ubiquitin Drives p97-Mediated Substrate Unfolding enhanced by FAF1, FAF2, and UBXD7
UFD1 Recognition of Initiator and Proximal Ubiquitin Drives p97-Mediated Substrate Unfolding enhanced by FAF1, FAF2, and UBXD7
Sato, Y.; Miyauchi, S.; Hirano, A.; Yasumoto, N.; Goto, Y.; Akizuki, Y.; Koyano, F.; Hino, T.; Nagano, S.; Matsuda, N.; Ohtake, F.; Hayashi, G.
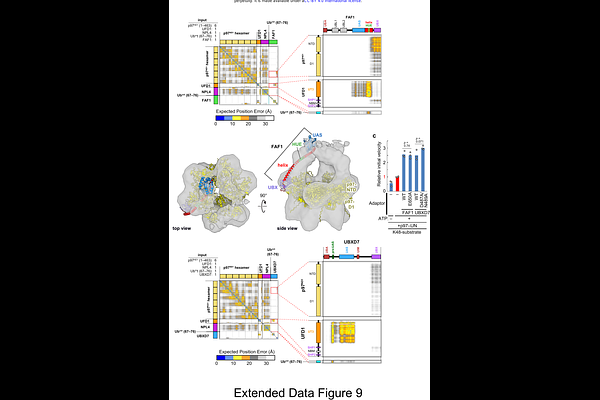
AbstractThe AAA-ATPase p97 and its primary adaptor UFD1-NPL4 (UN) unfold ubiquitinated substrates for their proteasomal degradation. Although cryo-EM has shown that substrate processing begins with unfolding of the initiator ubiquitin and its capture by NPL4, the specific contribution of UFD1 to K48-linked ubiquitin chain engagement has not been fully defined. Here we report a 1.31 [A] crystal structure of the human UFD1-UT3 bound to a K48-linked di-ubiquitin mimic, revealing that UFD1-UT3 engages the unfolded initiator ubiquitin via its Nc subdomain and the proximal ubiquitin via its Nn subdomain. AlphaFold3-guided analyses further demonstrate that accessory adaptors FAF1, FAF2, and UBXD7 enhance p97-UN activity through divergent strategies. FAF1 and FAF2 scaffold UFD1-UT3 near the NPL4-tower, whereas UBXD7 stabilizes transient interactions between UFD1 and the ubiquitin chain. Our results provide a comprehensive molecular model for the initiation of p97-mediated substrate unfolding.