KIF18A Inhibition as a Therapeutic Strategy in Cancers with Rb Pathway Inactivation
KIF18A Inhibition as a Therapeutic Strategy in Cancers with Rb Pathway Inactivation
Bakhoum, S. F.; Bowler, T.; Andreu, C.; Arora, A.; Chen, S.; Vedula, C.; Roopnariane, A.; Bettigole, S.; Bosco, N.; Dohadwala, A.; The SOVI-2302 Investigators, ; The VLS-1488-2201 Investigators, ; Southwell, D.; Ganem, N.
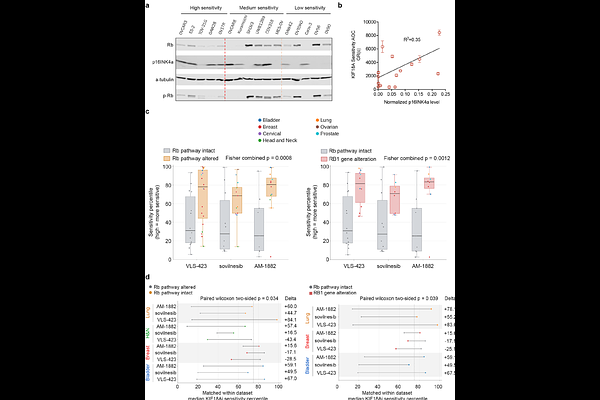
AbstractKIF18A inhibition has emerged as a therapeutic strategy for chromosomally unstable cancers, but clini-cal development is limited by the absence of a deployable predictive biomarker. Here we identify strong, diffuse p16INK4a expression, a well-established surrogate marker of Rb-pathway inactivation, as a predictive biomarker of response to KIF18A inhibition, and show that Rb-pathway inactivation marks a biologically distinct subset of cancers sensitive to this therapeutic approach. In sensitive models, low Rb activity is associated with robust spindle assembly checkpoint signaling and prolonged mitotic arrest fol-lowing KIF18A inhibition. Weakening the spindle assembly checkpoint in this context is sufficient to con-fer resistance. Across three independent pan-cancer sensitivity datasets generated with distinct KIF18A inhibitors, Rb-pathway altered models were significantly more sensitive than histology-matched Rb-intact comparators, with the strongest association observed in cancers harboring direct RB1 loss or in-activating mutation. Guided by this mechanism, we retrospectively analyzed p16INK4a expression by immunohistochemistry (IHC) in pre-treatment tumor biopsies from 79 heavily pre-treated high-grade serous ovarian cancer patients across three dose-escalation or expansion cohorts and treated with two different KIF18A inhibitors (sovilnesib and VLS-1488) sharing a common mechanism of action. p16INK4a-high tumors showed substantially higher objective response rates than their p16INK4a-low counterparts (36.0% versus 2.2%; P = 0.0002) and markedly longer progression-free survival (median 24.3 versus 7.9 weeks; hazard ratio, 0.16; P < 0.0001). These findings establish p16INK4a as a mecha-nistically-based, clinically implementable biomarker of clinical response to KIF18A inhibition that is poised to support pan-cancer development of KIF18A inhibitors guided by Rb-pathway inactivation.