Single-phage profiling illuminates viral individuality during cell fate determination
Single-phage profiling illuminates viral individuality during cell fate determination
Homaee, E.; Zhu, W.; Yao, T.; Golding, I.
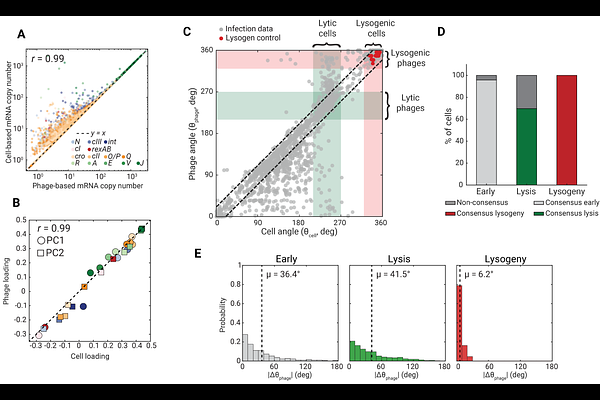
AbstractThe choice between cell death (lysis) and viral dormancy (lysogeny) following bacteriophage infection serves as a founding paradigm for the emergence of cellular heterogeneity in a genetically uniform population. The determination of host fate arises through the stochastic transcription from multiple viral genomes present within each cell, but this activity remains hidden from empirical interrogation, which typically stops at the whole-cell level. Here we use parallel sequential fluorescence in situ hybridization (par-seqFISH), followed by spatial clustering of phage-encoded transcripts within each cell, to profile the transcriptional activity of individual phages during synchronized infection of Escherichia coli (E. coli) by bacteriophage lambda. At the whole-cell level, transcription kinetics capture the developmental choice between lysis and lysogeny, and further demonstrate that viral replication is required for the emergence of diverging fate decisions. Zooming in to the single-phage level illuminates an individuality of viral activity during infection. We find that, while cells pursuing lysogeny display consensus activity of all inhabiting phages, lytic cells may contain phages that exhibit lysogenic activity. These findings support an earlier suggestion that consensus among coinfecting phages is required for cell dormancy. More broadly, our results highlight the need to identify how whole-cell behavior emerges from the activity of physically distinct copies of the same genetic circuit.