ULK1 drives NDP52-mediated selective autophagic degradation of MHC-I to promote immune evasion in HPV-positive head and neck cancer
ULK1 drives NDP52-mediated selective autophagic degradation of MHC-I to promote immune evasion in HPV-positive head and neck cancer
Vu, L.; Giacobbi, N. S.; Khalil, M. I.; Yang, C.; Eckerman, W. J.; Gomez Recinos, E.; Garber, J. D.; Son, H.; Chahal, P.; Villa, D. M.; Srivastava, T.; Bennett, A. Z.; Martin, K. R.; Welbon, C.; Williamson, C.; Spanos, W. C.; MacKeigan, J. P.; Olive, A. J.; Pyeon, D.
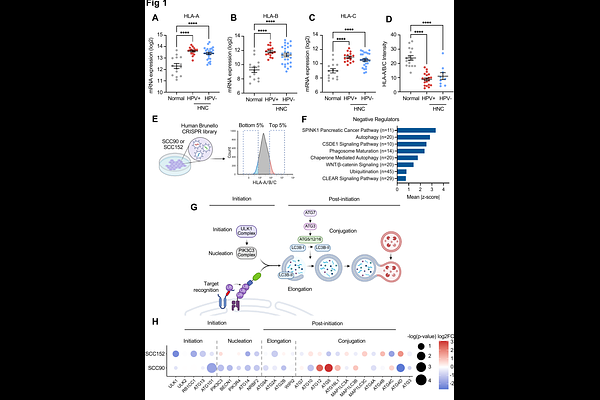
AbstractAntigen presentation by major histocompatibility complex class I (MHC-I) is critical for tumor cell killing by CD8+ T cells. Accordingly, tumor cells downregulate MHC-I expression through multiple mechanisms, thereby evading the immune response. Importantly, lower levels of MHC-I are associated with poor responses to immune checkpoint inhibitor therapy. Our recent study has shown that the human papillomavirus (HPV) oncoproteins induce MHC-I protein ubiquitination by membrane-associated Ring-CH-type finger 8 (MARCHF8) in HPV-positive head and neck cancer (HPV+ HNC). However, the mechanism by which ubiquitinated MHC-I is degraded remains elusive. By performing genome-wide CRISPR screens, we identified components of the ULK1 and PIK3C3 complexes for autophagy initiation complexes among the top negative regulators of cell-surface MHC-I expression in HPV+ HNC cells. We show that MHC-I is recruited from the ER to autophagosomes by the cargo receptor NDP52, decreasing MHC-I levels. Further, inhibiting the initiation or nucleation steps of autophagy before autophagosome formation is critical for restoring MHC-I levels on the cell surface. Finally, genetic inhibition of autophagy initiation suppresses HPV+ HNC tumor growth in vivo and enhances the CD8+ T cell-mediated antitumor response. Our findings suggest that autophagic degradation of MARCHF8-ubiquitinated MHC-I is a key immune evasion mechanism in HPV+ HNC.