Cell Cycle-Dependent Chromatin Motion: A Role for DNA Content Doubling Over Cohesion
Cell Cycle-Dependent Chromatin Motion: A Role for DNA Content Doubling Over Cohesion
Rey-Millet, M.; Costes, L.; Le-Floch, E.; Ayoub, H.; Saccomani, Q.; Manghi, M.; Bystricky, K.
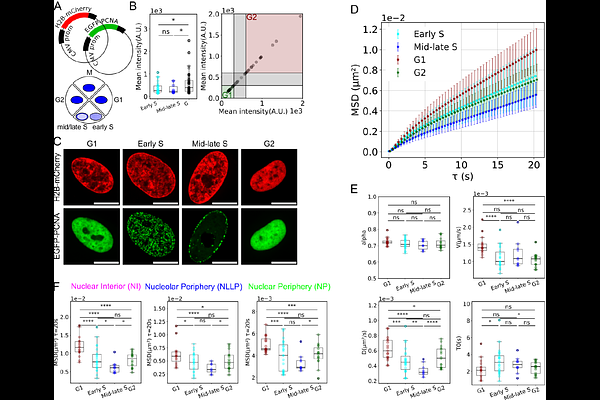
AbstractThe spatiotemporal organisation of chromatin in the eukaryotic nucleus is fundamental for genome regulation. Chromatin undergoes rapid remodelling and rearrangements within minutes, altering its diffusion properties. Considering the tight coupling between genome function and nuclear architecture, a key question is how chromatin dynamics adapt to or promote nuclear processes. To elucidate the underlying physical principles, we employed High-resolution Diffusion mapping (Hi-D) to track chromatin motion throughout interphase in live human cells. Our analysis, that considers both diffusive motion and drift generated by active forces, re-vealed that chromatin dynamics are heterogeneous, with distinct behaviours in different subnuclear zones. Notably, both diffusive and processive contributions to chromatin motion progressively decrease from G1 to G2 phase, with this reduction occurring uniformly across all subzones. This suggests a global mechanism driving the observed decrease in chroma-tin mobility during cell cycle progression. By combining genetic knockout experiments and polymer modelling, we demonstrate that the doubling of DNA content, rather than cohesin-mediated sister chromatid entrapment, is responsible for the gradual decrease in chromatin motion during the cell cycle in human nuclei. These findings provide new insights into the physical and functional organisation of chromatin and its regulation during cellular proliferation.