New three-dimensional preclinical models to understand and treat liver cancers activated for the β-catenin pathway
New three-dimensional preclinical models to understand and treat liver cancers activated for the β-catenin pathway
Bou Malham, V.; Leandre, F.; Hamimi, A.; Lagoutte, I.; Bouchet, S.; Gougelet, A.; Colnot, S.; Desbois-Mouthon, C.
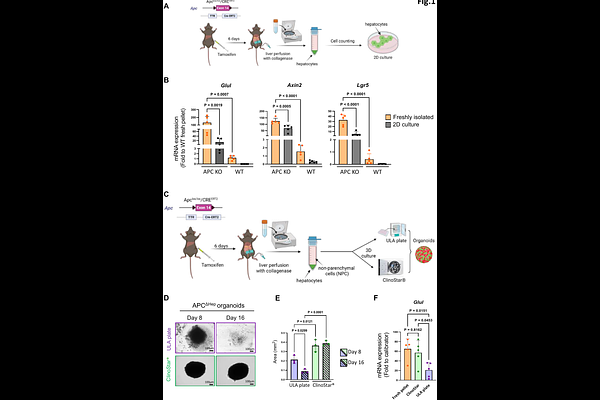
AbstractBackground & aims. Constitutive activation of the beta-catenin pathway is a determining feature in the pathogenesis of two primary liver cancers, namely HCC and hepatoblastoma (HB). Activating alterations in CTNNB1 gene and, to a lesser extent, inhibiting alterations in APC gene are observed in 30 to 40% of HCC cases and 80 to 90% of HB cases. For both tumours, therapeutic management is far from optimal. Therefore, relevant experimental models are needed to increase our knowledge and test new therapeutic approaches. Methods. Organoids and tumouroids were established from APCDhep and beta-catDex3 mouse models, which are clinically relevant models for beta-catenin-activated HCC and mesenchymal HB. We developed a new methodological approach based on a dynamic suspension culture in a rotating bioreactor. Morphological and molecular characteristics and sensitivity to WNTinib, a treatment already successfully tested on human HCC and HB tumouroids, were evaluated by histology, immunohistochemistry, immunofluorescence, and RT-qPCR. Results. This easy-to-implement methodology allows for the rapid generation of a large number of organoids and tumouroids that are uniform in size and show no signs of cell death in their core. The robustness of the methodology is illustrated by the maintenance of the histological architecture, cell diversity and gene expression in organoids and tumouroids in comparison with the native liver tissues. In addition, the value of the HCC-derived tumouroids for evaluating cancer treatment was assessed based on their responsiveness to the beta-catenin antagonist WNTinib. Conclusions. The organoids and tumouroids that we present here are new reliable in vitro cancer models, recapitulating the main features of beta-catenin-driven HCC and mesenchymal HB. They can be integrated into an appropriate platform for drug screening and could enable the development of a la carte therapies that are urgently needed for these indications.