Generative AI designs functional thiolation domains for reprogramming non-ribosomal peptide synthetases
Generative AI designs functional thiolation domains for reprogramming non-ribosomal peptide synthetases
Buelbuel, E. F.; Bang, S.; Geroge, K.; Bianchi, G.; Raj, P.; Chung, S.; Pauline, V.; Hochstrasser, R.; Minas, H. A.; Elgaher, W. A. M.; Kany, A. M.; Hirsch, A.; Schmitt, S.; Heinz, D. W.; Kalinina, O. V.; Klakow, D.; Bozhueyuek, K. A. J.
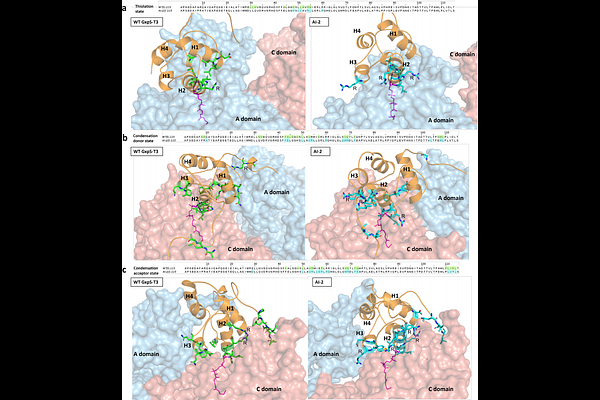
AbstractLarge language models and generative protein design promise to accelerate biotechnology, but it remains unclear whether they can engineer dynamic megasynth(et)ases whose activity depends on transient, context-specific domain interfaces. Nonribosomal peptide synthetases (NRPSs) are an especially demanding target, yet a high-value one because they produce many clinically important natural products and offer a route to analogs that are often difficult or impractical to access by chemical synthesis. Here we integrate pretrained generative models (ESM3, ProteinMPNN and EvoDiff) with design-build-test-learn cycles and data-guided prioritization to generate 76 de novo thiolation (T) domains. We built and tested 578 recombinant NRPS variants in vivo spanning minimal, full-length and hybrid assembly lines. AI-designed T-domains supported product formation across architectures, enabled catalytically active hybrids at recombined junctions and increased yields by up to ~3-fold relative to NRPSs carrying the native T-domain. A representative design showed improved soluble expression, refolding, and a 12 C higher melting temperature, while molecular dynamics simulations indicated preserved global stability but reshaped, state-dependent interdomain contact networks. Together, these results establish generative design as an effective route to context-conditioned optimization and reprogramming of biosynthetic assembly lines.