UBE3C retrofits the proteasome to enforce degradation of ultra-stable folds
UBE3C retrofits the proteasome to enforce degradation of ultra-stable folds
Zou, S.; Yin, D.; Shi, M.; Chen, E.; Luo, X.; Zhao, L.; Cao, S.; Wu, D.; Zhang, S.; Mao, Y.
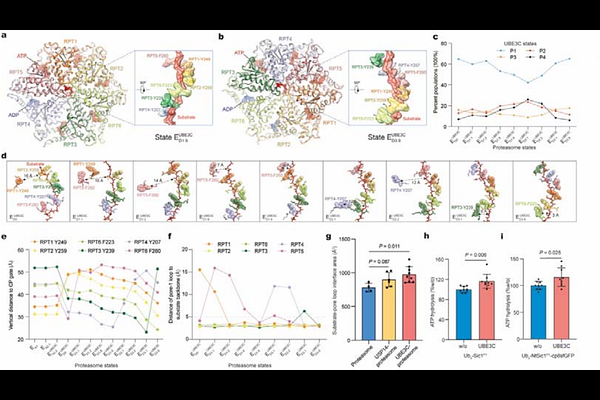
AbstractNeurodegeneration and proteinopathies arise from mutated or misfolded proteins that form ultra-stable, proteotoxic aggregates that evade proteasomal degradation despite ubiquitylation. These conditions are exacerbated by the deubiquitylase USP14 that prematurely rescues substrates. To counter these proteolysis-resistant effects, eukaryotes evolved the highly conserved HECT-type E3 ligase UBE3C, dysregulated in neurodegeneration and overexpressed in cancers, to reprogram the 26S proteasome toward heightened activity via unknown mechanisms. Here we visualized functional dynamics of the human UBE3C-retrofitted proteasome enforcing degradation of a re-engineered superfolder GFP that normally escapes proteolysis, using time-resolved cryo-electron microscopy. A continuum of non-equilibrium conformations, comprising fourteen proteasome conformers orthogonally combined with four UBE3C states or three USP14 states, reveals a cryptic E3-receptor site in the proteasomal lid and captures key intermediates of ubiquitin-chain elongation and branching at four linkage-specific ubiquitin-binding sites. Remarkably, UBE3C creates an extreme shortcut for ubiquitin shuttling that bypasses USP14, promotes USP14 recycling and allosterically strengthens substrate-unfolding forces of the AAA-ATPase unfoldase to ultimately surmount the unfolding energy barrier of ultra-stable proteins. These findings define the complete functional cycle of the UBE3C-reprogrammed proteasome, illuminating how UBE3C simultaneously controls USP14 and the proteasome to enforce clearance of ultra-stable folds, and establish crucial mechanistic foundations for proteolysis-targeting therapeutic discovery.