Membrane Proteome Remodeling in Female APP Mice Following Muscarinic Acetylcholine Receptor M1 Modulation Revealed by Peptidisc Enabled DIA-MS.
Membrane Proteome Remodeling in Female APP Mice Following Muscarinic Acetylcholine Receptor M1 Modulation Revealed by Peptidisc Enabled DIA-MS.
Duong van Hoa, F.
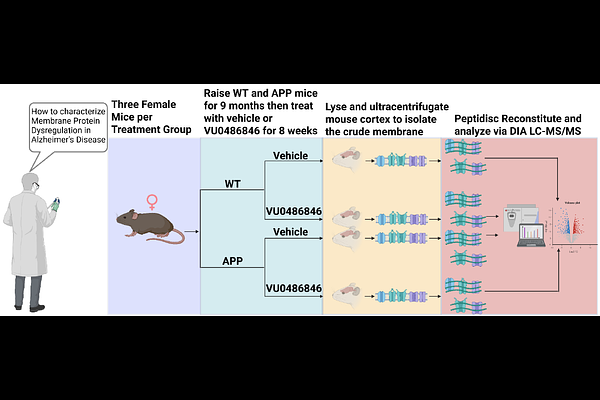
AbstractAlzheimer disease (AD) is linked to profound dysregulation of membrane-embedded and membrane-associated proteins that govern amyloid processing, synaptic signaling, and neuronal communication. Yet most proteomic analyses prioritize soluble fractions, resulting in systematic underrepresentation of integral membrane proteins and limited access to disease-relevant membrane pathways. Here, we use a membrane-mimetic, data-independent acquisition proteomic workflow to define disease- and drug-induced remodeling of the cortical membrane proteome in an APP mouse model of Alzheimer disease. Female B6C3F1/J mice were aged to 9 months and treated for 8 weeks with or without the M1 muscarinic acetylcholine receptor positive allosteric modulator VU0486846. APP pathology drove a pronounced, genotype-specific remodeling of the membrane proteome, with enrichment of multiple membrane proteins linked to AD, including RyR2, PLD3, ITM2C, and CNTNAP2. Wild-type mice cortical membranes were instead enriched for membrane proteins involved in axon guidance and synaptic organization, such as EPHA5 and ROBO2. In contrast, activation of M1 using the VU0486846 produced minimal membrane proteome changes in wild-type mice but selectively enriched proteins involved in neuronal trafficking and synaptic plasticity in APP mice, including SORCS2, PLXND1, and CADM1. Together, these findings demonstrate that AD-associated proteomic remodeling is strongly concentrated at the membrane level and that M1 receptor activation preferentially engages disease-altered membrane networks rather than inducing widespread proteomic changes. This work establishes peptidisc-enabled membrane proteomics as a powerful approach for identifying membrane-associated biomarkers and evaluating therapeutic target engagement in AD.