Perinuclear anchoring of telomeres enables plant infection by Ustilago maydis
Perinuclear anchoring of telomeres enables plant infection by Ustilago maydis
Sanz-Marti, E.; Velasco-Gomariz, M.; Navarrete, B.; Ibeas, J. I.; Barrales, R. R.
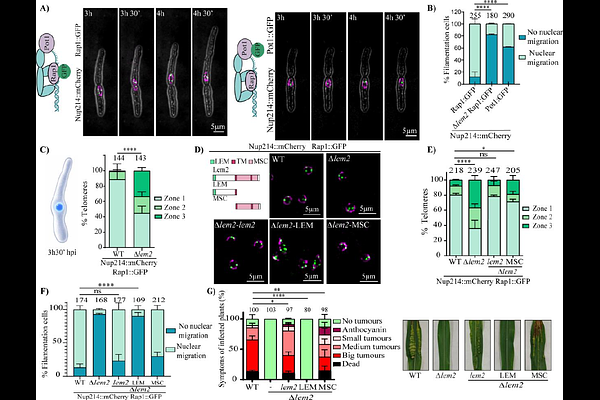
AbstractThe spatial organization of the genome is critical for cellular function, yet its contribution to the success of host-pathogen interactions remains poorly understood. A conserved hallmark of nuclear architecture is the perinuclear localization of heterochromatin, which includes not only specific chromosomal subdomains but also major structural elements like centromeres and telomeres. To determine the importance of this organization during fungal pathogenesis, we used the maize pathogen Ustilago maydis as an infection model system. We identify the lamina-associated protein homolog, Lem2, as a molecular tether for telomeres and show its deletion abrogates fungal penetration, causing developmental arrest at the appressorium stage. Mechanistically, this infection defect is associated with a failure in nuclear migration. Using hyphal filaments induced under axenic conditions, we observed that mutant nuclei consistently fail to transit from the mother cell into the growing filament. Furthermore, RNA-seq analysis correlates this failure with a marked misregulation of the DNA damage response (DDR) and cell cycle control, including the aberrant expression of multiple checkpoint and signaling proteins. Crucially, artificially tethering telomeres to the nuclear periphery in this mutant partially restores the nuclear migration defect and plant penetration, while simultaneously suppressing the aberrant DDR. This work establishes a functional link between a specific spatial nuclear configuration and fungal infectivity, revealing the conserved telomere-anchoring machinery as a potential target for novel antifungal strategies.