Adenylyl cyclase 9 as a molecular scaffold to dissect the regulatory mechanisms of membrane adenylyl cyclases
Adenylyl cyclase 9 as a molecular scaffold to dissect the regulatory mechanisms of membrane adenylyl cyclases
Kantarci, I.; Haoriwa, H.; Korkhov, V. M.
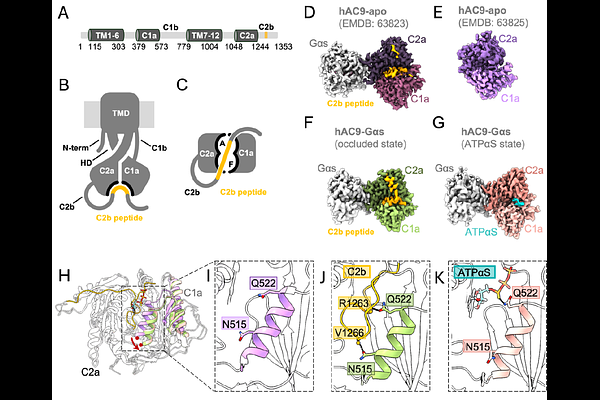
AbstractAdenylyl cyclases (ACs) convert ATP into the second messenger cAMP, thus directly influencing cellular signaling in response to a wide variety of stimuli. Despite their physiological importance, structural studies of isoform-specific AC regulation are compounded by difficulties in AC expression and purification. Here, we designed a chimeric construct AC95, combining human AC9 as a molecular scaffold and incorporating the catalytic-allosteric core of human AC5. Cryo-EM analysis of AC95 at 3.5 [A] resolution revealed a state of AC95 bound to both ATPS and forskolin, demonstrating that the chimera reproduces AC5-like allosteric regulation while retaining the structural features of the AC9 scaffold. Although AC95 chimera retained the ability to bind to and be activated by forskolin, it lost the ability to be autoinhibited by the C2b domain of AC9. Moreover, AC95 is insensitive to inhibition by specific AC5 inhibitors SQ22,536 and NKY80, suggesting that these molecules may target a site distinct from the catalytic-allosteric core of AC5 grafted into the AC95 chimeric construct. Our results establish a generalizable approach for investigating isoform-specific regulation of membrane ACs by small molecules, offering a potential path for structure-based drug discovery targeting distinct AC isoforms.