Fusion-driven post-transcriptional network orchestrates ferroptosis resistance, dormancy, and immune remodeling in PRCC-TFE3 renal cell carcinoma
Fusion-driven post-transcriptional network orchestrates ferroptosis resistance, dormancy, and immune remodeling in PRCC-TFE3 renal cell carcinoma
Mishra, D.; Agrawal, S.; Malik, D.; Pathak, E.; Mishra, R.
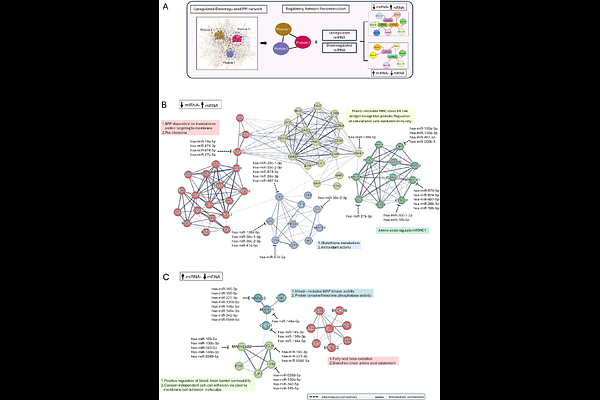
AbstractTFE3-rearranged papillary renal cell carcinoma (pRCC) remains poorly understood due to its rarity and complex biology. By integrating matched mRNA and miRNA sequencing from the TCGA-KIRP cohort, this study reveals a fusion-specific post-transcriptional program orchestrating ferroptosis resistance, metabolic adaptation, adhesion loss, immune remodeling, and dormancy-associated signaling. Fusion-positive tumors exhibited coordinated upregulation of antioxidant and mTORC1 components (SOD2, GPX4, G6PD, NQO1, FNIP2, RRAGC), reinforced by loss of miRNA-mediated repression. In parallel, elevated miRNAs (miR-185, miR-148a, miR-130b, miR-342) suppressed MAPK, Wnt, and tight-junction genes, generating a low-proliferative, adhesion-deficient state. AGO2-compatible miRNA-mRNA structural modeling validated key duplexes, linking miRNA regulatory specificity to fusion-driven transcriptional outputs. Immune profiling revealed reduced CD3 T cells but enrichment of CD8 and NK cells, supporting a remodeled yet persistence-permissive microenvironment. Finally, connectivity mapping (cMAP) analysis identified candidate leads targeting the MAPK-Wnt axis. These findings define a multi-layered RNA regulatory architecture unique to PRCC-TFE3 tumors.