A RAD18 SAP domain PIP motif enables PCNA mono-ubiquitination and USP1-BRCA1 synthetic lethality
A RAD18 SAP domain PIP motif enables PCNA mono-ubiquitination and USP1-BRCA1 synthetic lethality
Ashton, N. W.; Ravindranathan, R.; Korchak, E. J.; Somuncu, O. S.; Zambrano, G. A.; Asada, S.; Korzhnev, D.; Bezsonova, I.; D'Andrea, A. D.
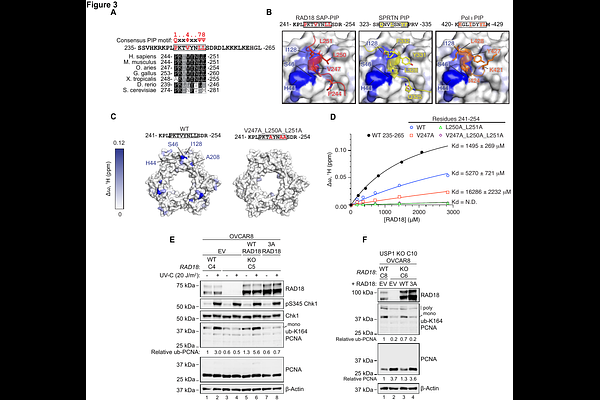
AbstractThe proliferating cell nuclear antigen (PCNA) sliding clamp is a central component of eukaryotic DNA synthesis. In response to DNA damage, PCNA is mono-ubiquitinated by the RAD6 (E2)-RAD18 (E3) complex. However, the structural basis by which RAD18 engages PCNA to direct mono-ubiquitination has remained poorly defined. Mono-ubiquitinated PCNA can subsequently be extended with K48-linked poly-ubiquitin chains that target PCNA for degradation. Ubiquitin-specific protease 1 (USP1) reverses both mono- and poly-ubiquitination of PCNA; accordingly, USP1 inhibition promotes accumulation of mono-ubiquitinated PCNA at replication forks and reduces total PCNA levels, leading to replication defects that are synthetically lethal with BRCA1 deficiency. Here, we combine computational and structural approaches to identify and characterize a SAP domain PCNA-interacting peptide (PIP) motif within RAD18. We demonstrate that this interaction is required for DNA damage-induced PCNA mono-ubiquitination and for PCNA turnover following USP1 loss. Disruption of the RAD18-PCNA interface suppresses ssDNA gap accumulation and reduces USP1 inhibitor sensitivity in BRCA1-deficient cells. Furthermore, cells adapted to prolonged USP1 inhibition exhibit reduced RAD18 levels, suggesting that deregulation of PCNA mono-ubiquitination represents a biologically relevant resistance mechanism. Together, these findings define a structural interface required for RAD18-dependent PCNA mono-ubiquitination and establish it as a key determinant of USP1-BRCA1 synthetic lethality.