Kinetic proofreading as a mechanism for transcriptional specificity in living human cells
Kinetic proofreading as a mechanism for transcriptional specificity in living human cells
Kim, J. M.; Ball, D. A.; Johnson, T. A.; DInzeo, C.; Cho, H. J.; Ozbun, L.; Karpova, T. S.; Pegoraro, G.; Larson, D. R.
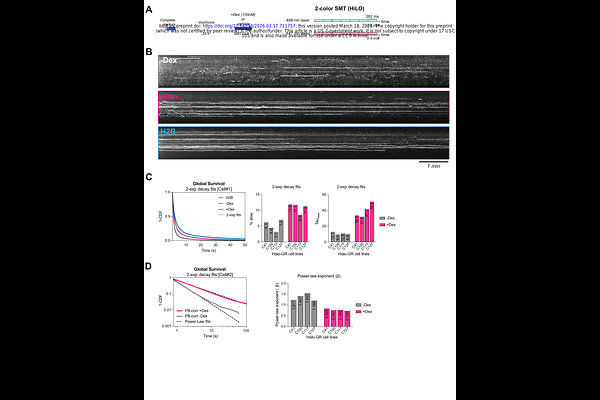
AbstractHow target genes selectively respond to their specific transcription factors (TF) amid the vast excess of non-specific TFs in the nucleus remains a fundamental question in gene regulation. Here, we develop an integrated single-molecule imaging framework to quantitatively link TF dynamics with nascent transcription kinetics at endogenous gene loci to address the transcription factor specificity. Using endogenously Halo-tagged glucocorticoid receptor (GR), we show that ligand activation rapidly increases GR chromatin binding and residence times without substantially altering search kinetics. Live-cell nascent RNA imaging reveals that GR is essential for Dex induction of ERRFI1 transcription via increased burst frequency. Gene locus-specific, dual-color tracking demonstrates that GR display longer residence times in general near its target gene ERRFI1 compared to the non-target MYH9 locus, consistent with a kinetic proofreading model. A high-throughput imaging-based CRISPR screen identifies ATP-dependent processes, including neddylation and chromatin remodeling, and similarly, acute inhibition of the TFIIH XPB selectively impairs ERRFI1 transcription while sparing MYH9, implicating ATPase activity in GR target discrimination. In all, these findings establish that promoters function as dwell-time rather than occupancy detectors, discriminating specific from non-specific TF interactions through energy-dependent mechanisms.