OKN4395, a first-in-class EP2/EP4/DP1 triple antagonist reprograms prostanoid-driven immunosuppression to restore antitumor immunity
OKN4395, a first-in-class EP2/EP4/DP1 triple antagonist reprograms prostanoid-driven immunosuppression to restore antitumor immunity
Grandclaudon, M.; Boulch, M.; Thaller, A.; Sabio-Ortiz, J.; Grimaldi, A.; Goxe, M.; Knopf, A.; Daugan, M. V.; Huehn, E.; Gnerre, C.; Jeay, S.; Faronato, M.; Dakhli, H.; Lopez-Lastra, S.; Hardy, A.; Sanchez, S.; Mayer, I.; Hoste, R.; Montanari, F.; Soumelis, V.; Alberti, J.; Pattarini, L.; Hoffmann, C.; Pierce, A. J.
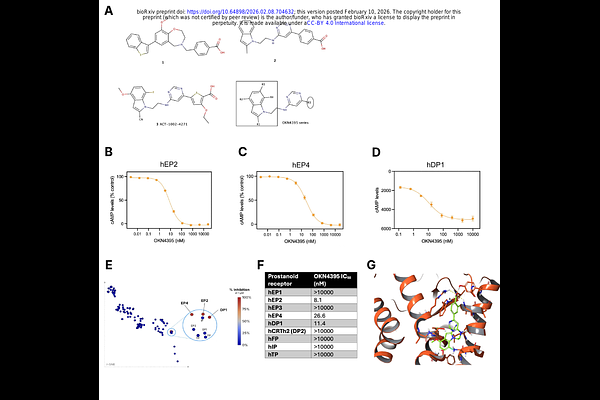
AbstractImmune checkpoint inhibitors, particularly T cell targeting anti-PD(L)1 therapies, have revolutionised the treatment landscape for solid malignancies, but challenges related to non-responsiveness and the development of treatment resistance continue to be observed. An additional immunosuppressive axis relates to prostaglandin signaling downstream of cyclooxygenase-2 (COX2), where COX2 inhibitors have shown clinical promise in re-engaging both T and non-T cell immune compartments, yet have suffered from toxicity concerns. We report here the preclinical characterization of OKN4395, a highly potent and specific first-in-class triple antagonist of EP2, EP4, and DP1, major tumor immunosuppressive receptors downstream of COX2. OKN4395 restores immune function on both T cells and NK cells in vitro. Additionally, OKN4395 acts synergistically with anti-PD1 to increase speed and depth of antitumor activity. Overall, these findings robustly support the clinical investigation of OKN4395 in an ongoing Phase 1 trial (NCT06789172) as an innovative cancer immunotherapy for solid tumors, as a single agent and in combination with anti-PD1 therapy.