Mechanical evolution of 3T3 fibroblastic cells exposed to nanovibrational stimulation
Mechanical evolution of 3T3 fibroblastic cells exposed to nanovibrational stimulation
Johnson-Love, O.; Espinosa, F. M.; Tejedor, J. R.; Gorgone, G.; Campsie, P.; Dalby, M.; Reid, S.; Garcia, R.; Childs, P.
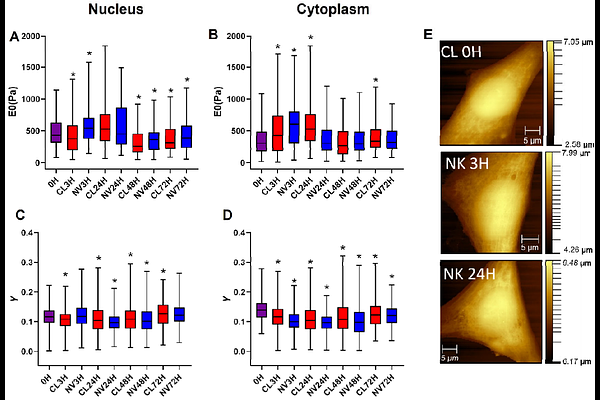
AbstractCells are mechanosensitive, responding to external mechanical stimulation. Nanovibrational stimulation has been shown to enhance cell contractility and actin stress fibre formation. These changes in morphology occur quickly, alongside associated mechanical changes. Here, the relationship between acute morphological and mechanical changes in NIH 3T3 fibroblastic cells in response to nanovibrational stimulation is presented. A 1 kHz, 30 nm vibration is applied continuously for 72 hours. Atomic force microscopy (AFM) quantifies mechanical properties of the nucleus and cytoplasm at multiple timepoints, while immunofluorescence tracks morphological changes. Within 3 hours of stimulation, both nuclear and cytoplasmic stiffness increase significantly, accompanied by a decrease in the cellular fluid exponent, suggesting a shift of the cell towards more solid-like behaviour. These changes correlate with increased nuclear area. Actin polymerisation also increases within 24 hours, although variably. To understand the role of the cytoskeleton, actin polymerisation and contraction are inhibited using cytochalasin D and blebbistatin. Results show that inhibition prevents stiffness increases and results in a higher fluid exponent, indicating a more fluid-like state. These findings demonstrate that actin-myosin dynamics mediate cell stiffening under nanovibrational stimulation. Interestingly, prolonged stimulation appears to reverse this effect, suggesting that temporal optimisation of stimulation may enhance long-term mechanotransducive responses.