The role of N-glycans and their processing in ER-to-lysosome-associated degradation of disease-causing mutant Neuroserpin
The role of N-glycans and their processing in ER-to-lysosome-associated degradation of disease-causing mutant Neuroserpin
Fregno, I.; Hoefner, C.; Molinari, M.
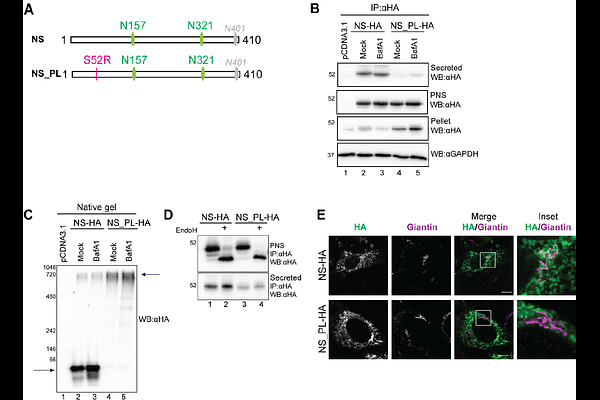
AbstractMost proteins synthesized in the endoplasmic reticulum (ER) are covalently modified upon addition of pre-assembled oligosaccharides to side chains of asparagine (N) residues. Processing of N-linked oligosaccharides by ER-resident glucosidases, mannosidases and glucosyltransferases determines the fate of the associated polypeptides. Terminally glucose residues are removed from N-glycans to hamper engagement of ER-resident glucose-binding chaperones and promote secretion of native polypeptides. Mannose residues are removed to target terminally misfolded proteins for dislocation across the ER membrane and clearance by the cytoplasmic ubiquitin proteasome system (ER-associated degradation, ERAD). Recent evidence highlights the role of persistent N-glycan glucosylation as a signal that promotes segregation of misfolded proteins in ER subdomains that are eventually delivered to endolysosomal compartments for ER-to-Lysosome-Associated Degradation (ERLAD). Here we show that the polymerization-prone Portland variant of Neuroserpin (NS_PL) associated with familial encephalopathy with NS inclusion bodies (FENIB) is a client of the ERLAD machinery. Its lysosomal clearance relies on the LC3-dependent delivery branch of ERLAD involving the lectin chaperone Calnexin (CNX), the ERphagy receptor FAM134B and the SNARE protein Syntaxin17 (STX17), which is engaged upon persistent glucosylation of the NS_PL oligosaccharide linked at the asparagine residue at position 321.