Astrocytes modulate mitochondrial proteostasis and membrane potential in neurons through S100A6 - calcyclin-binding protein signaling
Astrocytes modulate mitochondrial proteostasis and membrane potential in neurons through S100A6 - calcyclin-binding protein signaling
Cinquina, V.; Keimpema, E.; Alpar, A.; Verkhratsky, A.; Harkany, T.
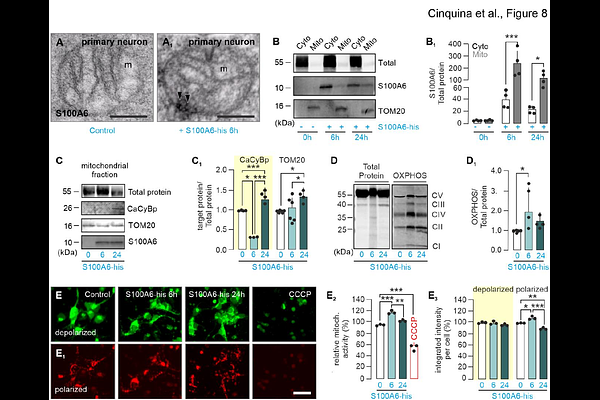
AbstractCalcyclin binding protein (CaCyBp) is an evolutionarily conserved protein that converts S100 protein family-derived signals to protein turnover through an ubiquitin ligase interaction domain within its C-terminus. Despite its predicted roles in cellular differentiation, CaCyBp expression alone and in relation to S100A6, its prototypic ligand, during fetal and postnatal organ development remains incompletely understood. Here, we combined cell-resolved neuroanatomy and biochemistry to reveal not only organ system-level CaCyBp expression but also its region-, circuit-, and neuron-specific distribution in the mouse brain from embryonic day 18.5. We extended these studies to human brains past gestational week 27, corroborating that neurons, but not astrocytes, microglia or oligodendrocytes, harbored CaCyBp. Conversely, S100A6 was detected only in astrocytes and ependymoglia in both species. Ultrastructural studies localized CaCyBp to mitochondria in neurons, with genetic loss-of-function and recombinant S100A6-induced signaling implicating CaCyBp in the regulation of the mitochondrial electrochemical gradient controlling ATP production. Overall, pleiotropic CaCyBp expression in neurons suggests a maintenance receptor-like role impacting cellular respiration, thus affecting proteostasis upon glia-derived S100A6 signals.