POLQ-driven repair scars shape the immunogenic landscape of homologous recombination-deficient pancreatic cancer
POLQ-driven repair scars shape the immunogenic landscape of homologous recombination-deficient pancreatic cancer
Park, W.; Umeda, S.; Hilmi, M.; O'Connor, C. A.; Sharma, R.; Tezcan, N.; Zhang, H.; Zhu, Y.; Schwartz, C.; Yaqubie, A.; Varghese, A. M.; Soares, K.; Florou, V.; Kim, D.; Maron, S.; Argiles, G.; Balogun, F.; McIntyre, C.; Kim, D.; Yu, K. H.; Chou, J. F.; Hayashi, A.; Keane, F.; Khalil, D. N.; Chatila, W. K.; Capanu, M.; Chaligne, R.; Pishvaian, M. J.; Bandlamudi, C.; Lecomte, N.; Berger, M.; Basturk, O.; Balachandran, V.; Pe'er, D.; Rousseau, B.; Greenbaum, B.; Sfeir, A.; Iacobuzio-Donahue, C. A.; Riaz, N.; O'Reilly, E. M.
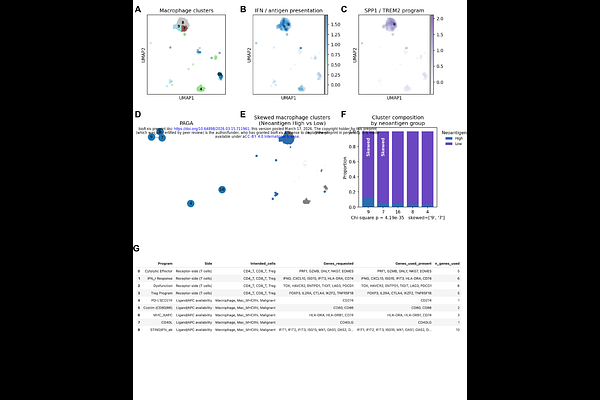
AbstractPancreatic cancer (PC) is broadly resistant to immune checkpoint blockade, although a subset of homologous recombination-deficient (HRD) tumors exhibits durable immune engagement. The genomic features that distinguish these immune-responsive tumors from immune-inert HRD tumors remain poorly understood. Here we identify a microhomology-mediated end joining (MMEJ) repair scar, the MMEJ Deletion Footprint (MDF), as a genomic readout of POLQ-associated error-prone repair that enriches for frameshift indels. Across the multi-omic discovery cohort integrating tumor genomics, single-nucleus transcriptomics and spatial immune profiling, MDF-high HRD PC exhibited increased frameshift-indel-derived neoantigens and interferon programs. MDF was further associated with remodeling of the myeloid compartment toward MHC II-high dendritic cell-like antigen-presenting macrophage states and the immune synapse architecture marked by increased spatial interaction between APC-like macrophages and cytotoxic CD8+ T cells. These tissue-level features aligned with a functional trajectory shift of CD8+ T cells, consistent with effective anti-tumor immunity and was associated with favorable clinical outcomes of patients. Together, our findings position MMEJ-linked repair scarring as actionable biology that connects an HRD genotype to immune organization and suggests rational immunotherapy combinations that may enhance antigen presentation and myeloid activation to extend durable benefit in HRD-lineage cancers.