Pioneer factor IRF1 unlocks latent enhancers to rewire chromatin and immunometabolism in inflammatory macrophages
Pioneer factor IRF1 unlocks latent enhancers to rewire chromatin and immunometabolism in inflammatory macrophages
Ayala, J.-M.; Bellworthy, R.; Mancini, M.; Ibarra-Meneses, A. V.; Fernandez-Prada, C.; Langlais, D.
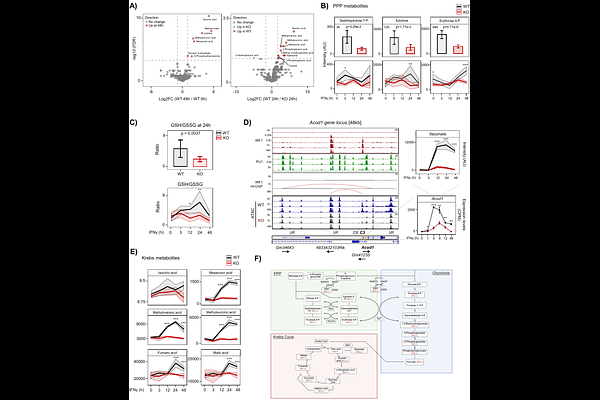
AbstractMacrophages undergo extensive chromatin and metabolic remodeling to mount effective inflammatory responses. Here, we identify Interferon Regulatory Factor 1 (IRF1) as a pioneer factor that orchestrates these processes during IFN{gamma}-driven macrophage activation. Integrative ATAC-seq, ChIP-seq, Hi-ChIP, and nascent RNA-seq demonstrated that IRF1 rapidly engages closed chromatin, initiates enhancer remodeling, and drives removal of repressive histone marks followed by deposition of activating modifications. IRF1-established enhancers form long-range chromatin interactions with target promoters, activating transcriptional programs that control immune effector functions, chromatin regulation, and cellular metabolism. Notably, IRF1 coordinates the IFN{gamma}-induced metabolic switch from oxidative phosphorylation to aerobic glycolysis by transcriptionally regulating key metabolic enzymes, with metabolite profiles consistent with increased glycolysis and pentose phosphate pathway engagement, while remodeling the tricarboxylic acid (TCA) cycle to support immunometabolic outputs. IRF1-deficient macrophages fail to execute this coordinated metabolic reprogramming. High-density IRF1 motif arrays promote enhanced chromatin occupancy and recruitment of the BRG1-containing chromatin remodeling SWI/SNF complex. Pharmacologic inhibition of SWI/SNF ATPase activity (SMARCA2/4) disrupts IRF1-dependent chromatin remodeling and gene induction. Moreover, IRF1-induced enhancer states persist after IFN{gamma} withdrawal, establishing durable epigenetic memory. Together, these findings establish IRF1 as a central integrator of chromatin remodeling, transcriptional control, and metabolic adaptation, linking pioneer-factor activity to macrophage plasticity and innate immune memory.