Integrative AlphaFold Modeling, Fragment Mapping, and Microsecond Molecular Dynamics Reveal Ligand-Specific Structural Plasticity at the Human Urotensin II Receptor
Integrative AlphaFold Modeling, Fragment Mapping, and Microsecond Molecular Dynamics Reveal Ligand-Specific Structural Plasticity at the Human Urotensin II Receptor
Torbey, A. G.
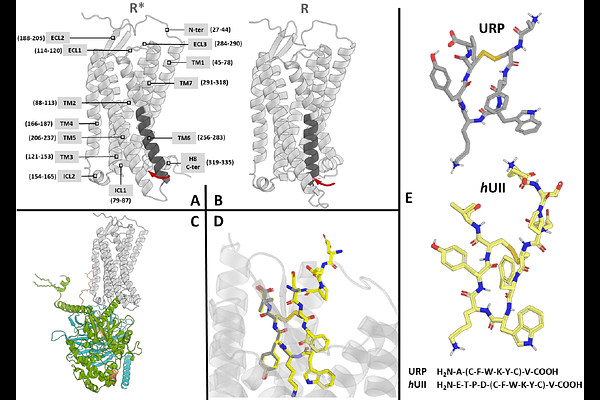
AbstractPeptide ligands Urotensin II (hUII, human), hUII-related peptide (URP) and its cognate human receptor (hUT) are known for their implications in cardiovascular pathophysiology, yet the lack of experimentally resolved hUT structures has limited a deep mechanistic understanding of ligand binding and receptor activation. Here, we leverage recent breakthroughs in multistate AlphaFold predictions, long-timescale molecular dynamics (MD) simulations, and site identification by ligand competitive saturation (SILCS) based pocket mapping and solving ligand bound conformation to illuminate the dynamic interaction of hUII and URP with hUTR. By analyzing hUT dynamics in its intracellular transducer binding pocket, and residue-level interaction probabilities in each simulation, we capture subtle distinctions in the way hUII and URP anchor key pocket residues, modulate transmembrane (TM) domain tilts. Results indicate that hUII imposes stronger conformational constraints on TM5 and TM6 relative to URP, both potentially stabilizing different active-like receptor configurations. At the same time, interaction maps highlight unique aromatic and polar networks that each ligand exploits. These findings reinforce the concept that relatively small differences in GPCR peptide ligand structure may lead to large effects on receptor-state selection, signal specificity, ultimately reflecting different clinical outcomes. By integrating computational modeling with per-residue dynamics, this work not only reconciles prior mutagenesis and docking data but also provides validated 3D models and MD simulations of the endogenous ligands bound to hUT, offering new opportunities to selectively harness ligand-dependent signaling in the urotensinergic system.