Discovery of Novel Ligands for Cryptococcus neoformans
Discovery of Novel Ligands for Cryptococcus neoformans
Benfeito, S.; Alves, C.; Lima, C.; Borges, F.; Sequeira, L.; Cagide, F.; Rocha, T.
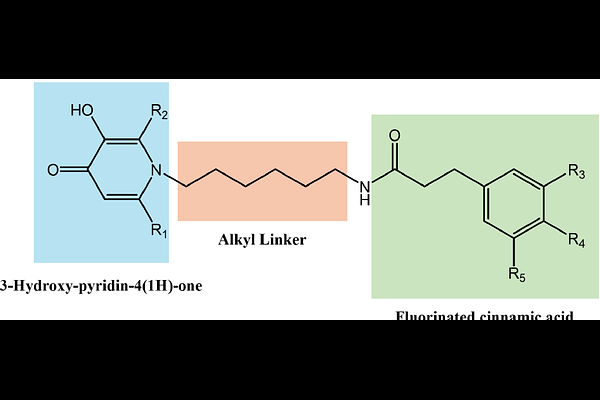
AbstractFungal pathogens are an escalating global public health concern, particularly in the context of invasive and opportunistic infections. Cryptococcosis, primarily caused by Cryptococcus neoformans var. grubii, can manifest as acute, subacute, or chronic disease, affecting multiple organs and frequently leading to life-threatening meningitis in immunocompromised individuals. Given the limited antifungal therapeutic strategies and the emergence of resistance and toxicity-related constraints, the development of novel anti-cryptococcal agents remains an urgent priority. In this study, a library of innovative hybrids (5a to f) based on the 3hydroxypyridin4(1H)one scaffold was developed. Their antimicrobial activity was evaluated towards a panel of clinically relevant Gram-positive (methicillin-resistant Staphylococcus aureus: MRSA) and Gram-negative bacteria (Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Acinetobacter baumannii), as well as fungal species Candida albicans and Cryptococcus neoformans var. grubbi. Cytotoxicity was assessed in HEK293 and HepG2 cell lines, and haemolytic profile was determined to evaluate safety. In addition, iron-chelating capacity and lipophilic properties were also investigated. All compounds formed stable complexes with iron(III) and were non-toxic at concentrations up to 25M. Lipophilicity studies showed that compounds in series 1 (5a to c) exhibited lower lipophilicity than those in Series 2 (5d to f), mainly due to the regioisomeric position of the hydroxyl group on the 2-methyl-4-pyridone scaffold; specifically, the C3substitution pattern in Series 2 that enhances the hydrophobic character compared to the C5substitution in Series 1. Fluorination further increased lipophilicity in both series. Notably, compounds 5c to 5f emerged as potent, selective, and non-toxic antifungal agents against Cryptococcus neoformans var. grubii (MIC < 16 g/mL; CC50 > 32 g/mL; HC10 > 32 g/mL). Their distinct structural features appear to play a key role in antifungal selectivity, supporting the potential of these 3-hydroxypyridin4(1H)one-based hybrids as promising approach for the development of novel therapeutics for cryptococcal meningitis.