Magnesium as a Conformational Gatekeeper of KRAS: Structural Dynamics and Therapeutic Implications
Magnesium as a Conformational Gatekeeper of KRAS: Structural Dynamics and Therapeutic Implications
Srinivasu, B. Y.; Damerla, T. S.; Stec, A.; Zhou, Z.; Engen, J. R.; Westover, K. d.; Wales, T. E.
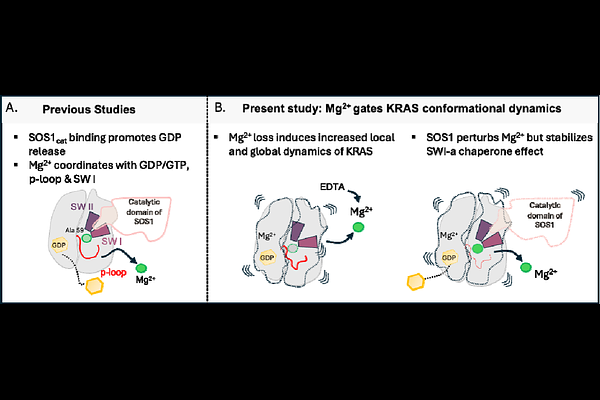
AbstractMagnesium serves as an essential cofactor for small GTPases, yet its structural role in regulating KRAS conformational dynamics and nucleotide exchange remains poorly understood. Here, we combine hydrogen deuterium exchange mass spectrometry (HDX-MS), native mass spectrometry, and functional assays to elucidate how Mg2+ stabilizes the KRAS conformational ensemble and constrains transitions between GDP- and GTP-bound states. Depletion of Mg2+ triggers widespread increases in structural dynamics throughout KRAS, spanning the p-loop, 1-helix, switch I, nucleotide-binding region, and distal helices, revealing a global loosening of the protein fold that favors an open, nucleotide exchange-competent state. Mg2+ titration experiments demonstrate that individual structural elements exhibit distinct Mg2+ dependencies: the p-loop and 1-helix recover native dynamics at micromolar concentrations, whereas switch I requires millimolar levels, underscoring its exceptionally high sensitivity to Mg2+ for structural stabilization. KRAS bound to the catalytic domain of exchange factor SOS1 displays an HDX signature closely resembling the Mg2+-free state, indicating that SOS1 promotes nucleotide exchange by transiently perturbing Mg2+ coordination while simultaneously stabilizing switch I. Consistently, phosphomimetic KRAS S17E variant, which disrupts a critical Mg2+-coordinating residue, exhibits pronounced global destabilization, reinforcing the central importance of Mg2+ in maintaining structural integrity. Taken together our findings show that Mg2+ acts as a master regulator of KRAS structural dynamics and reveal Mg2+-sensitive hotspots that might represent promising targets for next-generation KRAS therapeutics.