NF-κB transcriptionally enhances p53 accumulation dynamics hampering DNA repair
NF-κB transcriptionally enhances p53 accumulation dynamics hampering DNA repair
Colombo, E.; Pozzi, S.; Loffreda, A.; Genova, F.; Aloi, E.; Heinichen, T.; Falletta, P.; Mazzocca, M.; Fillot, T.; Gnani, D.; Agresti, A.; Bianchi, M. E.; Zambrano, S.; Mazza, D.
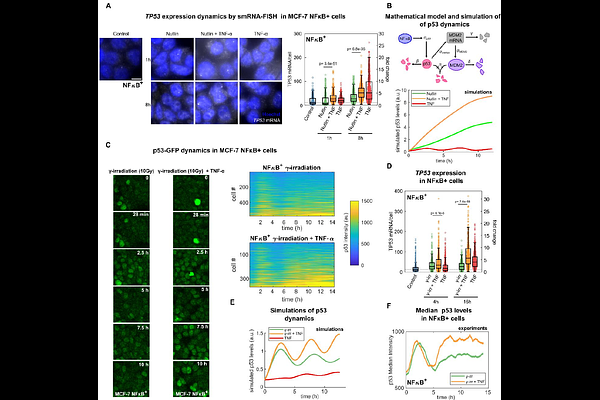
AbstractCells integrate multiple, often concurrent signals through intertwined genetic circuits whose dynamics shape transcriptional programs and cell fate decisions. Among these, the tumor suppressor p53 and the inflammatory transcription factor NF-{kappa}B are central regulators of stress responses in normal and cancer cells, yet their dynamic crosstalk under co-activation remains poorly characterized. Here, we combine genetic approaches, live cell imaging, transcriptomic analysis and mathematical modeling to dissect their dynamic interplay. We find that co-activation of NF-{kappa}B by the inflammatory cytokines TNF- and IL-1{beta} significantly enhance p53 nuclear accumulation upon genotoxic stress or Nutlin3a, and this effect is absent in NF-{kappa}B-deficient cells. Mechanistically, we show that cytokines induce an NF-{kappa}B-mediated increase of TP53 transcription, and mathematical modeling indicates that it is sufficient to account for the observed increased p53 accumulation. Functionally, NF-{kappa}B co-activation rewires p53-dependent transcriptional programs and impairs p53-mediated DNA repair following genotoxic stress, due to a shift of p53 dynamics from oscillatory to more sustained accumulation; p53 oscillatory dynamics and DNA repair remain largely unaltered in absence of NF-{kappa}B. Our results uncover an amplification of p53 response in presence of inflammatory cues that is transcriptionally mediated by NF-{kappa}B and that results, counterintuitively, in functional antagonism.