Endocrine-metabolic decoupling drives stress vulnerability in dystrophin deficiency
Endocrine-metabolic decoupling drives stress vulnerability in dystrophin deficiency
Major, G. S.; Timpani, C. A.; Lalunio, H.; Chen, J.; Boatner, L.; Giourmas, N.; Kourakis, S.; van den Berg, E.; Salimova, E.; Eliades, J.; Zheng, G.; Xu, R.; Bhangu, S. K.; Mendis, T. V. H.; Merry, D. L.; Lantier, L.; Hagemeyer, C. E.; Cavalieri, F.; Caeyenberghs, K.; Russell, A. P.; de Veer, M.; Goodman, C. A.; Hayes, A.; Rybalka, E.; Najt, C. P.; Lindsay, A.
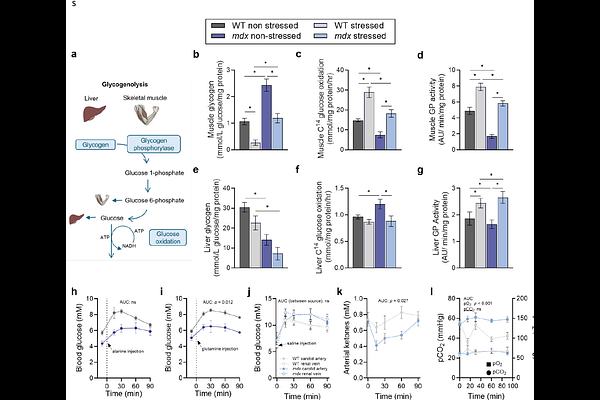
AbstractSkeletal muscle orchestrates systemic metabolism, dynamically coordinating glucose uptake and fuel use to match energy demand. In Duchenne muscular dystrophy, loss of dystrophin derails this control, exposing a hidden metabolic weakness. In the mdx mouse, we show that physiological stress exposes a primary failure in insulin-stress axis integration: unchecked glucocorticoid signalling outpaces insulin secretion, driving systemic hyperglycaemia despite preserved muscle insulin sensitivity. These data support multi-tissue dystrophinopathy driving endocrine-metabolic decoupling. Skeletal muscle glycogen accumulates excessively and resists mobilisation under stress. The heart maintains high glucose uptake, whereas the brain remains glucose-limited, defining tissue-specific vulnerabilities. Acute insulin supplementation normalises systemic glucose uptake and rescues stress-induced behavioural deficits. Likewise, empagliflozin-mediated glucose offloading stabilises blood glucose and enhances muscle function to levels comparable with standard care prednisolone. These findings identify endocrine-metabolic decoupling as a central driver of metabolic fragility in DMD, correctable though insulin restoration or targeted glucose redirection.