Localization-dependent activation of the DEAD-box ATPase Vasa by eLOTUS domains
Localization-dependent activation of the DEAD-box ATPase Vasa by eLOTUS domains
Garbelyanski, A.; Hauser, D.; Trolese, A.; Kumar, P.; Gan, H. H.; de Almeida, C.; Meyer, M. P.; Hondele, M.; Carlomagno, T.; Gunsalus, K. C.; Jeske, M.
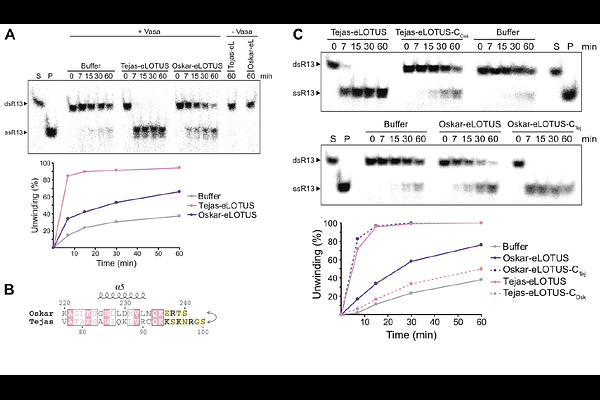
AbstractDEAD-box RNA helicases remodel RNA structures in many cellular pathways, yet how their activity is spatially controlled in cells remains poorly understood. The Drosophila germline helicase Vasa functions in ovaries only when localized to cytoplasmic granules, a process mediated by eLOTUS-domain proteins. Here, we define the mechanism by which eLOTUS domains activate Vasa. Biochemical analyses reveal that Vasa alone is largely inactive. eLOTUS domains bind the open conformation of Vasa and promote formation of the closed RNA- and ATP-bound state by accelerating RNA engagement. This stimulation requires a positively charged intrinsically disordered sequence within eLOTUS that increases RNA association. Mutations in this element abolish Vasa stimulation while preserving binding. We demonstrate that Vasa activation is essential in vivo. Together, these findings reveal a localization-dependent mechanism for regulating the DEAD-box helicase Vasa, in which enzymatic activity is gated by a spatially restricted cofactor.