A conserved archaeal ribosome-associated factor linking bacterial hibernation and eukaryotic energy sensing
A conserved archaeal ribosome-associated factor linking bacterial hibernation and eukaryotic energy sensing
Souza, D. P.; May, M. B.; Carrion, J.; Alva, V.; Bisson, A.; Davis, J. H.
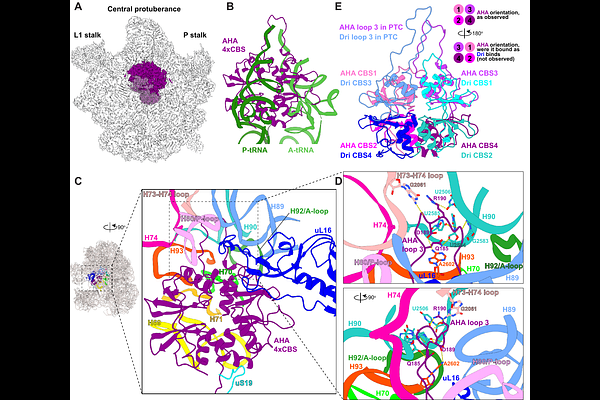
AbstractRibosome hibernation helps cells survive stress by reversibly silencing translation and limiting degradation of ribosomal complexes. Although well characterized in bacteria and eukaryotes, archaeal hibernation remains poorly understood. Using cryoEM to analyze lysates from a model archaeon, we identified AHA (AMPK{gamma}-HPF from Archaea), a broadly conserved ribosome-associated protein factor composed of two distinct modules. Structural analyses showed that AHA's C-terminal domain binds the small subunit, while its N-terminal region recognizes the large subunit, occluding the mRNA channel and the A- and P-tRNA binding sites and thereby enforcing translational silencing. Consistent with this proposed function, {Delta}AHA cells displayed reduced viability, depletion of ribosomal proteins during stationary phase, and impaired recovery upon return to growth. Phylogenetic analyses revealed that AHA's C-terminal domain shares homology with the bacterial Hibernation Promoting Factor (HPF), indicating an origin in the last universal common ancestor (LUCA) and thereby identifying HPF as a universal hibernation module. Strikingly, we observed two AMP molecues bound to AHA's N-terminal CBS-tetrad, which we found was structurally and evolutionary related to the eukaryotic energy sensor AMPK{gamma}, thus linking energy sensing between archaea and eukaryotes. Together, these findings uncover a widespread archaeal ribosome hibernation factor and establish a direct evolutionary link between prokaryotic translational silencing and eukaryotic energy sensing.