Castration-resistant prostate cancer cells are addicted to the high activity of cyclin-dependent kinase 2
Castration-resistant prostate cancer cells are addicted to the high activity of cyclin-dependent kinase 2
Chatterjee, J.; Marin, A.; Yalala, S.; Itkonen, H. M.
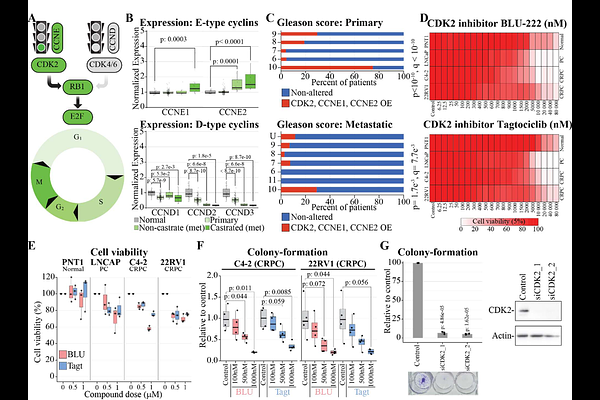
AbstractBackground Cyclin-dependent kinases drive the progression through the cell cycle and thereby form classical targets for cancer therapy. In prostate cancer (PC), the first line of therapy typically targets androgen receptor (AR), but it frequently leads to development of incurable form of the disease, castration-resistant PC (CRPC). Here, we sought to understand if CRPC cells are selectively addicted to a specific cell cycle kinase. Methods We used PC and CRPC patient data to evaluate transcriptional changes and modeled the responses in vitro using multiple models of PC, CRPC and normal cells. Development of a CDK2 inhibitor-resistant CRPC cell line, and a compound screen were used to identify chronic and acute vulnerabilities to augment the efficacy of our candidate therapy in multiple PC, CRPC and also normal cells, to assure selectivity. Results We show that the emergence of CRPC is associated with significant upregulation of cyclins that positively regulate cyclin-dependent kinase 2 (CDK2) and downregulation of CDK4 cyclins. Accordingly, CDK2-specific inhibitors and its knock down efficiently reduce proliferation of PC and CRPC cells. CDK2 inhibitor-resistant CRPC model displayed transcriptional rewiring of cell cycle regulators, characterized by a shift towards CDK4/6-dependency and increased AR-signaling. Combinatorial drug screen discovered both antagonistic and additive combinations, and we show that AR inhibitors selectively augment the efficacy of CDK2 inhibitors against PC and CRPC cells, but the combination is not toxic to normal cells. Conclusion We discovered that CRPC cells are addicted to high CDK2 activity and show that combination of CDK2 inhibitors with the currently used anti-CRPC therapies selectively augment their efficacy.