LC-MS profiling of prmt-1 and prmt-5 knockout C. elegans reveals PRMT-1 substrates and global proteome remodeling
LC-MS profiling of prmt-1 and prmt-5 knockout C. elegans reveals PRMT-1 substrates and global proteome remodeling
Basirattalab, A.; Wallis, D. C.; Hartel, N. G.; Jalalifarahani, M.; Phillips, C. M.; Graham, N. A.
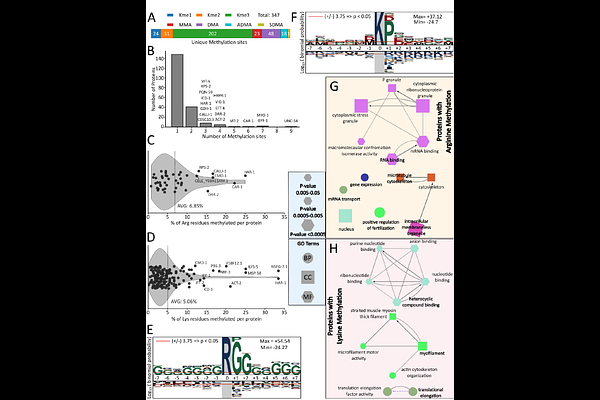
AbstractAlthough protein arginine methylation regulates diverse biological processes, it remains understudied relative to other post-translational modifications. Here, we analyzed C. elegans prmt-1 and prmt-5 null mutants using LC-MS proteomics to map PRMT methylation substrates and to quantify the effects of PRMT knockout on global protein abundance. High-pH strong cation exchange fractionation was used to enrich methylated peptides, and parallel analysis of whole cell lysates was used to measure global protein abundance. Quantitative methyl-proteomics identified 31 PRMT-1-dependent methyl-arginine peptides from 15 proteins with several arginine residues demonstrating dramatic decrease in both monomethyl- and asymmetric dimethyl-arginine abundance. Whole-proteome profiling revealed that prmt-1 knockout caused broad remodeling of the worm proteome with changes linked to DNA replication/cell-cycle programs, protein folding, and amino acid metabolism. Although prmt-5 knockout affected similar biological pathways to prmt-1 knockout, the effects on the C. elegans proteome were more modest. Together, these data connect PRMT-dependent methylation changes to proteome remodeling in a whole-animal model, support previous work suggesting that PRMT-1 is the dominant Type I PRMT in C. elegans, and provide a resource for studying how PRMT-1 and PRMT-5 shape protein regulation in vivo. All raw data have been deposited in the PRIDE database with accession number PXD074042.