Metabolic maintenance of breast cancer cells and metastases throughE-cadherin/YAP-dependent pyruvate carboxylase expression
Metabolic maintenance of breast cancer cells and metastases throughE-cadherin/YAP-dependent pyruvate carboxylase expression
Balamurugan, K.; Weiss, J. M.; Sharan, S.; McKennett, L.; Donohue, D.; McVicar, D. W.; Sterneck, E.
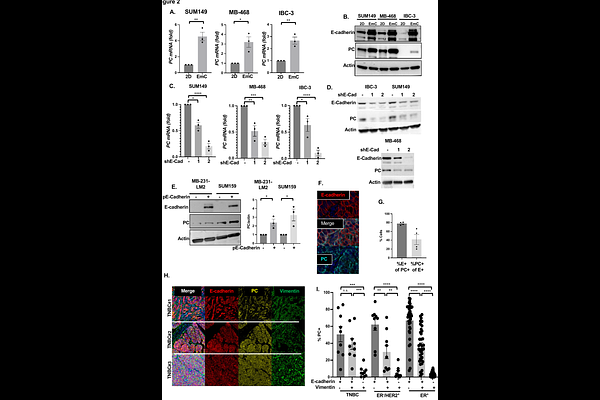
AbstractEpithelial-mesenchymal transition (EMT) and glycolytic metabolism are well-characterized drivers of cancer progression and metastasis. However, most primary breast tumors and metastases express E-cadherin and the epithelial phenotype is associated with mitochondrial oxidative metabolism, yet the causality and relevance of these relationships and their underlying mechanisms remain poorly understood. Using a 3D culture model with mechano-stimulation, we found that E-cadherin promotes mitochondrial oxidative phosphorylation (OXPHOS) while reducing oxidative stress. Through pharmacological and genetic manipulations of inflammatory breast cancer (IBC) and/or triple negative breast cancer (TNBC) cell lines, we identified pyruvate carboxylase (PC) as an E-cadherin effector. Critically, restoring PC in E-cadherin-silenced cells rescued mitochondrial oxygen consumption and protection from oxidative stress. Co-expression of E-cadherin and PC was confirmed in breast cancer tissues and experimental lung metastases. Mechanistically, E-cadherin induced PC expression and OXPHOS via AKT-mediated activation of YAP/ /TEAD transcription factors, which are better known as supporting EMT. Clinically relevant AKT and TEAD inhibitors reduced both PC expression and oxidative respiration. Importantly, PC inhibition as monotherapy attenuated or reduced established experimental lung metastasis burden in mice. These findings reveal that E-cadherin-mediated cell-cell adhesions directly support mitochondrial metabolism through AKT-YAP/TEAD-PC signaling, identifying a therapeutic vulnerability in metastatic epithelial TNBC.