Flow molecular dynamics simulations reveal mechanosensitive regulation of von Willebrand factor through glycan-modulated autoinhibitory modules
Flow molecular dynamics simulations reveal mechanosensitive regulation of von Willebrand factor through glycan-modulated autoinhibitory modules
Richard Louis, N. E. L.; Zhao, Y. C.; Ju, L. A.
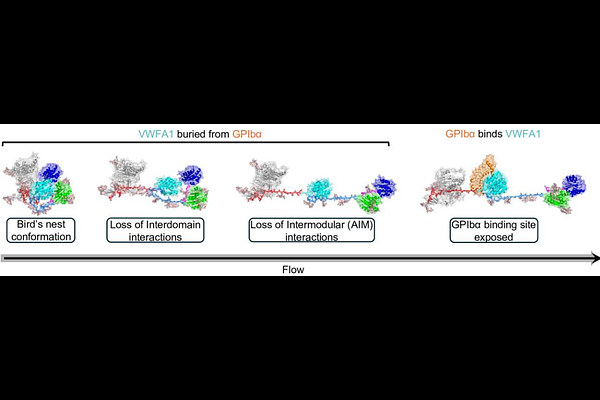
AbstractForce-induced protein conformational changes govern many essential biological processes, yet their molecular mechanisms remain difficult to resolve. Von Willebrand factor (VWF), a central regulator of haemostasis, is activated by hydrodynamic forces in blood flow, but how mechanical signals propagate across its multidomain architecture is poorly understood. Here, we use flow molecular dynamics (FMD), a simulation framework that applies fluid forces via controlled solvent flow to interrogate mechanosensitive proteins. Using VWF as a model system, we reconstructed the complete mechanomodule (DD3A1A2A3; 1,109 residues) with native glycosylation by integrating crystallographic data and AlphaFold predictions. FMD simulations capture a force-driven transition from a compact, autoinhibited bird-nest ensemble to an extended, activated state, revealing asymmetric autoinhibitory strengths within the NAIM and CAIM modules of the A1 domain. By directly linking static structures to dynamic, force-regulated behaviour, this work establishes a generalizable platform for dissecting protein mechanosensitivity and enabling the rational design of force-responsive therapeutics.