Structural variants in human congenital heart disease disrupt distal genomic regulatory contacts of developmental genes
Structural variants in human congenital heart disease disrupt distal genomic regulatory contacts of developmental genes
Lee, J.; Wu, J.; Pittman, M.; Grant, Z.; Kuang, S.; Quait, D.; Morton, S.; Fudenberg, G.; Traglia, M.; Hayes, K.; Pediatric Cardiac Genomics Consortium, ; Kumar, R.; Bruneau, B.; Pollard, K. S.
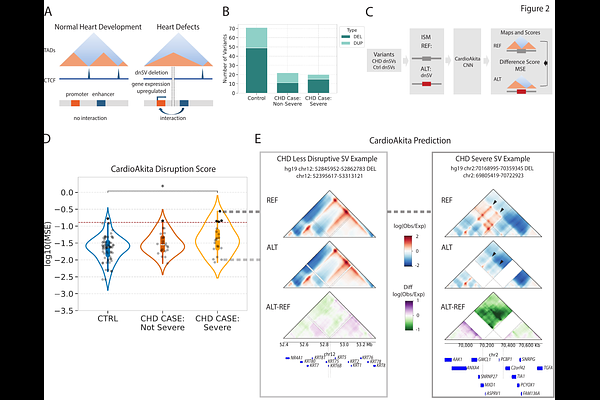
AbstractPredicting the functional significance of structural variants (SVs) associated with genetic diseases remains challenging. To test the hypothesis the SVs from people with congenital heart disease (CHD) disrupt developmental chromatin interactions, we developed CardioAkita, a machine-learning model that predicts how variants alter 3D chromatin structure. Analyzing previously genotyped de novo SVs (dnSVs), we observed a positive association between CHD severity and CardioAkita scores across dozens of families. From whole-genome sequencing of three individuals with CHD we predicted disruptive dnSVs. Induced pluripotent stem cells engineered to harbor these variants confirmed CardioAkita's predictions of 3D chromatin changes, and further revealed aberrant expression of local genes including cardiac developmental genes, suggesting that chromatin reorganization plays a significant mechanistic role in the genetic etiology of CHD. Our findings highlight the potential for models of 3D chromatin organization to predict the pathogenicity and underlying mechanisms of SVs in human disease.