FDA-approved drug library screen identifies antidepressants, antimicrobials, anti-COPD, and anti-CVD agents as blockers of NLRP3 inflammasome and sepsis in a sex-dependent manner.
FDA-approved drug library screen identifies antidepressants, antimicrobials, anti-COPD, and anti-CVD agents as blockers of NLRP3 inflammasome and sepsis in a sex-dependent manner.
Timinski, K.; Neupane, K.; Prince, A.; Bhandari, N.; Khan, M.; Sharma, S.; Shiravand, Y.; Traughber, C. A.; Raquepaw, Z.; Gulshan, K.
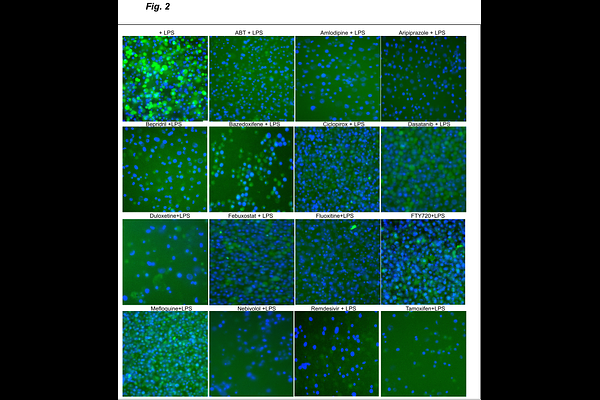
AbstractThe NLRP3 inflammasome pathway is central to host defense, but dysregulated activation of inflammasomes promotes diseases associated with metabolic syndrome (diabetes, obesity, CVD, MASLD), neurodegenerative diseases (Alzheimer's and Parkinson's), autoinflammatory conditions (CAPS, gout), and respiratory illnesses (asthma/COPD, and COVID-19). Therapeutic modulation of NLRP3 is challenging as it requires selective blockade of detrimental inflammasome activation without broadly suppressing innate immunity. Here, we used a phenotypic screen in THP 1 ASC GFP monocytes to identify FDA-approved drugs that can block LPS-induced priming of NLRP3 inflammasome or inhibit NLRP3 assembly (ASC speck formation) without disrupting upstream priming. Various classes of drugs, such as antidepressants (Fluoxetine, Duloxetine), antihypertensives (Irbesartan, amlodipine, nebivolol), antidiabetics (Rosiglitazone), {beta}-adrenergic agonists (Salmeterol), antimalarials (Mefloquine), antifungals (Azoles, ciclopirox), and antivirals (Saquinavir, Remdesivir), were identified as potent blockers of either priming or assembly of NLRP3 inflammasome. Hits were validated in several biochemical assays, including effect on release of proinflammatory cytokines, autophagy, lysosomal biogenesis, LPS binding, NF-kB nuclear localization, mitochondrial membrane potential, mitochondrial ROS, and biophysical properties of the cell membrane. A subset of identified drugs was tested in murine studies to probe effects on NLRP3 inflammasome assembly/activation and LPS-induced sepsis. Mice treated with ASC puncta blockers showed markedly reduced proinflammatory cytokines in peritoneal lavage and plasma. Mice treated with LPS-priming blockers showed a sex-specific increase in survival rate in the mouse model of LPS-induced mortality, validating the in vitro screen. Further studies in primary human cells and in vivo disease models are needed to assess the repurposing and therapeutic relevance of identified drugs.