Extracellular Vesicle-Enriched Secretome from Mesenchymal Stromal Cells Protects Against Chemically, Particulate-, and Ischemia-Induced Innate-Immunity Induced Inflammation
Extracellular Vesicle-Enriched Secretome from Mesenchymal Stromal Cells Protects Against Chemically, Particulate-, and Ischemia-Induced Innate-Immunity Induced Inflammation
Park, K.-s.; Ordouzadeh, N.; Lazzari, L.; Elia, N.; Scarpitta, S.; Iachini, M. C.; Bussolati, B.; Bruno, S.; Grange, C.; Ceccotti, E.; Prudente, D.; Cedrino, M.; Di Bucchianico, S.; Ryffel, B.; Quesniaux, V.; Togbe, D.; Huaux, F.; Wilmot, J.; Lallo, E.; Lotvall, J.; Dominici, M.
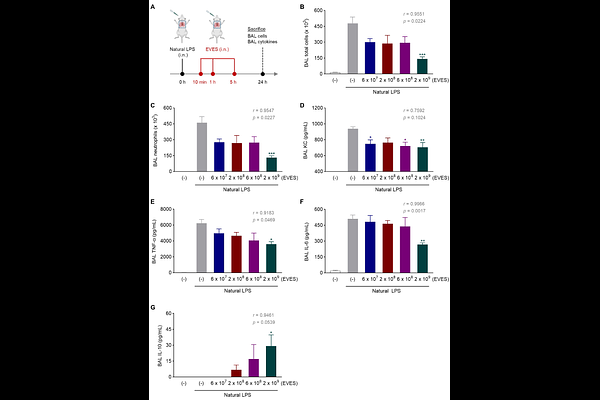
AbstractMesenchymal stromal cells (MSCs) are multipotent cells with well-established regenerative and immunomodulatory properties, making them promising candidates for the treatment of inflammatory diseases. However, the therapeutic effects of MSCs are largely mediated by their secretome, particularly extracellular vesicles (EVs), which deliver bioactive molecules capable of modulating inflammatory responses. We generated an extracellular vesicle-enriched secretome (EVES) from MSCs under scalable, Good Manufacturing Practice (GMP)-compliant conditions and assessed its therapeutic efficacy in diverse disease models, including lung inflammation and kidney injury induced by distinct innate immune stimuli. EVES was isolated from the secretome of umbilical cord blood-derived MSCs cultured in a chemically defined medium. In vitro, EVES significantly and dose-dependently attenuated cytokine release from airway epithelial cells and macrophages stimulated with inflammatory agents such as lipopolysaccharide or reactive particles. In murine models of lung inflammation, EVES reduced neutrophil infiltration and suppressed multiple cytokines and chemokines in a dose-dependent manner. In models of kidney injury, EVES enhanced tubular epithelial cell proliferation, improved renal histology, and markedly reduced tubular necrosis following ischemia-reperfusion injury. Collectively, these findings demonstrate that MSC-derived EVES exhibits robust and broad-spectrum therapeutic activity across multiple disease contexts driven by innate immune activation, supporting its potential as a scalable, cell-free therapeutic platform.