Short-Term Performance Assay Identifies Functional Benefits and Early Toxicity of Longevity Interventions in Mice
Short-Term Performance Assay Identifies Functional Benefits and Early Toxicity of Longevity Interventions in Mice
Marin-Jerez, E.; Rueda-Carrasco, J.; Melendez-Rodriguez, F.; Partido-Borge, P.; Tapia, E.; Leibowitz, B. D.; Parras, A.
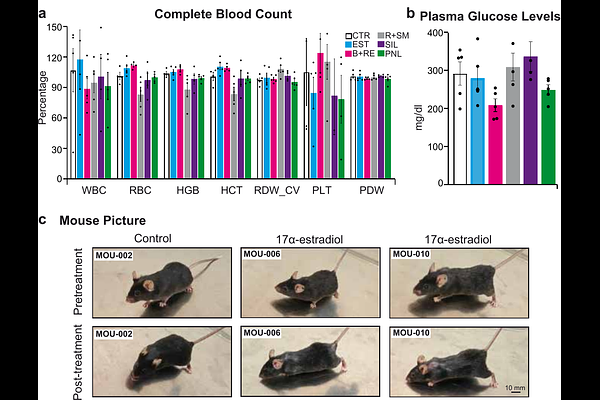
AbstractMouse lifespan studies are slow and costly, limiting the number of interventions that have demonstrated robust anti-aging effects. This highlights the need for rapid early-stage screening tools capable of assessing both efficacy and potential side effects. Here, we present a short-term performance assay designed to rapidly profile functional benefits and early toxicity of longevity interventions in mice. Over an 8-week period, mice received one of five candidate anti-aging treatments: 17a-estradiol, rapamycin + Smer28, berberine + resveratrol, sildenafil and pinealon. The protocol longitudinally monitored body weight and temperature, and food intake, alongside post-treatment assessments of grip strength, locomotor activity, Y-maze cognition, social behavior, and hematological and urinary parameters. The screen revealed compound-specific phenotypes: 17a-estradiol induced significant weight loss, increased grip strength, and dorsal alopecia, consistent with metabolic remodeling. Sildenafil reduced basal body temperature and preserved locomotor activity. Berberine + resveratrol decreased food intake and fasting glucose without major changes in physical performance, resembling caloric restriction-like metabolic effects. Rapamycin + Smer28 modestly improved strength and sociability but induced anemia in 2 of 5 mice, indicating potential dose-dependent toxicity. Pinealon showed a trend toward improved working memory without detectable adverse effects. This multi-parametric approach enables discover healthspan extending interventions facilitating prioritization and dose refinement before committing to full lifespan studies. Finally, to our knowledge, this represents the first comprehensive preclinical aging study in mice fully funded through tokenized decentralized science (DeSci), demonstrating how community-governed, on-chain funding can support resource-intensive in vivo research.